Tuberculosis: tackling the troubling uptick
Before Covid, the bacterial infection “tuberculosis” was the number 1 infectious disease killer on the planet. Every day it claims the lives of thousands, with the impact particularly marked in lower income countries which account for 80% of the cases. But that doesn’t mean that richer countries are off the hook. As people migrate, and wars displace refugees, Western countries are seeing a rising trend in new TB cases, particularly of highly drug-resistant forms of the infection. So this week we’re looking at new ways to combat one of the oldest and most fearsome infections on the planet…
Risa Bagwandin, who conceived and compiled this programme, was kindly supported by an award from AfricaBio, and a foundation run by Nick Draeger, that enabled her to join the Naked Scientists for 2 months in 2022 to receive special training in science communcation. We think she's done a wonderful job and clearly learned a great deal during her time with us. Thank you Risa, and thank you to AfricaBio.
In this episode
What is tuberculosis?
Sebastian Lucas
Risa Bagwandin went to meet pathologist and world-renowned TB expert, Sebastian Lucas, surrounded by specimens from people who have fallen victim to TB at King’s College London’s Gordon Museum…
Sebastian - Tuberculosis is an infection caused by a bacterium called mycobacterium tuberculosis. It's a very common infection and we think about 10 million people a year globally fall ill from it, and it has a relatively high mortality. It has been with us for tens of thousands of years. Most people catch it by being breathed over by people who have got active tuberculosis, who are excreting the bacteria from their lungs. So every time they breathe out, tens, thousands, millions of tubercle bacilli come out and they're breathed in by another person and it goes into their lungs and starts the process of TB infection.
Risa - What are the symptoms if someone does contract tuberculosis?
Sebastian - It's estimated that if a hundred people get infected, 10 will develop disease. And the standard dogma is that half of those will get it within the first five years and the other half might get it later on in their life. The practical point is that a lot of people get infected by tuberculosis and they will have lesions in their lungs and in most cases those lesions will not progress. They will be evident on a chest x-ray later, but those people will not have actual disease. The small proportion of people who do develop active disease have an infection focus in the lungs, which enlarges and produces a mass, maybe the size of one's thumb or a little bit bigger, and the tubercle bacilli will then spread to the local lymph nodes and from there they may well spread through any other part of the body.
Risa - What are the first signs of someone that has tuberculosis?
Sebastian - It depends how severe the disease is. The majority of people acquiring tuberculosis for the first time will not know they've actually had it. I, for one, was infected around the early 1980s, but that only became evident when someone did a chest x-ray some years later and said, 'Oh, you've had old TB there'. And I said, 'Really? I was never ill'. And that's fairly typical, but if one is ill, the primary features are going to be cough. And then also the systemic features, if the infection spreads, of weight loss and just feeling ill. Coughing up blood comes a lot later, but basically cough that does not resolve is a very good clue that someone might have tuberculosis and if they have weight loss as well, that's a very good clue.
Risa - The initial symptoms are very similar to other diseases. For example, it could be confused with a common seasonal flu or bacteria that cause pneumonia. This makes it quite difficult for diagnosis to occur.
Sebastian - Absolutely. To prove tuberculosis properly, you have to see or identify the organisms. That can be done in a coughed up sputum and you can stain it and look under the microscope and see the tubercle bacilli. If a person therefore is symptomatic and they have demonstrated bacteria, then that is an absolutely classic definite case of tuberculosis. But a lot of people can have lung tuberculosis and one is unable to prove the organism because it is simply not there in large enough numbers to identify. The second thing is imaging chest x-rays and other sorts of chest imaging are very important in identifying tuberculosis. They will not prove that a lesion is tb, but there will be supportive evidence and as a result, certainly in high income countries, a lot of people will be treated as if they did have tuberculosis, but they may not because it was never provable.
Risa - In that case, if they have TB and we still haven't proved it, it means that they're also capable of transmitting it unknowingly.
Sebastian - That's an interesting question: Who transmits tuberculosis? We've been talking so far about people who have acquired tuberculosis for the first time, primary tuberculosis. Now primary tuberculosis does not generally destroy large areas of the lung. This point is important because if the lung is being destroyed, it generally means that you have an abscess cavity. Air now gets into those cavities and tubercle bacilli love growing in air, so there's a sudden very large rate of producing organisms that you can cough out. The practical point is that younger people don't produce these cavities. It's people who have tuberculosis generally later in life who have cavitated. They are the people who infect everyone else.
Risa - We're surrounded by specimens of previous TB patients. Can you give us a description of how disease progress in the body to actually cause the destruction that we see on the specimens?
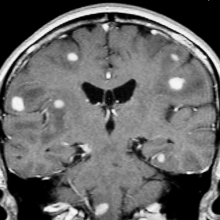
Sebastian - Well, let's first look at this one, which is a lung slice. You can see a two centimeter white mass that is a tuberculoma, a massive caseating tuberculosis. This is a primary infection, so that's been breathed in and has produced the tubical focus there. It's spread a bit locally. It will also spread to the lymph nodes and here one can see from the lymph nodes that it's actually spread to other parts of the body because the rest of the lung has got these little tiny white spots. This is what we call miliary tuberculosis, Little tiny seeds of tuberculosis, which has come through the bloodstream via the lymph nodes. And this similar specimen here, which is a spleen from a child, is covered in little white miliary spots. So that child has had primary tuberculosis in the lung. It hasn't healed and is actually spread to other parts of the body. Kidney, liver, and bone marrow. Another place that tuberculosis can go to very readily is the brain. And here they've gone to the brain covering the meninges. These are the membranes around the brain and the spinal cord. And if you look at the base of the brain there, it looks rather murky because it's covered in an exudate of custardy material, which is tuberculous inflammation. You can see the whole spinal cord is covered in yellow, orangey material and that is a very severe example of tuberculous meningitis. The next specimen, we're looking at a lung here at which the upper part is completely destroyed. And you see these cavities, the important thing is the tub basc inside these cavities grow like mad, go into the bronch eye and being coughed up all the time. So that person with lungs like that until he died was chronically infecting other people all the time.
Risa - Is it possible that without being treated people actually die of tuberculosis?
Sebastian - In these cases, yes, because we know the dates when the specimens were acquired, which was before effective anti TB therapy came in, which is just after the second World war. And so these are historical examples of untreated tuberculosis. And the patient with the tuberculous meningitis divide of acute hydrocephalus, the patient with a cavitating tuberculosis here died of lung failure and the child with the splenic miliary tuberculosis, given that that will be affecting all other parts of his immune system, could well have died of immune failure. Now with therapy, you stop all that, the spleen can go back sort of to normal. The brain, the inflammation would certainly decrease and you may be able to rescue the patient. If you look at the destroyed cavitated lung TB therapy is not going to give you new lung tissue because it doesn't regrow. But if the damage has not been so severe, then that person can carry on living with, albeit reduced, lung function, but is still alive.
09:21 - TB or not TB? That is the question.
TB or not TB? That is the question.
Keertan Dheda, University of Cape Town & London School of Hygiene and Tropical Medicine
These treatments are all we have to tackle TB once someone has become diseased due to the lack of an effective vaccine. As a consequence, the control and elimination of TB is highly dependent on rapid diagnosis. According to the WHO, 1.6 million people died from TB in 2021, many of which will have been due to late or missed diagnosis preventing prompt treatment. Professor Keertan Dheda jointly from the University of Cape Town and The London School of Hygiene and Tropical Medicine.
Keertan - As has already been outlined, the diagnosis of TB is a huge problem. 4 million roughly of the 11 million newly ill people with TB every year, or roughly one in three patients with active TB, remain undiagnosed or undetected. And this is a startling statistic. And there are three key reasons why the diagnosis of TB is problematic or, put in another way, the weak links in the chain are threefold. Firstly, the public health or casefinding strategy for reasons of affordability is inappropriate. And by this I mean patients are expected to self report when they have symptoms. And we call this passive case finding. And this is in contradistinction to active case finding where healthcare workers or diagnostics are taken out of the laboratories and healthcare facilities into the community. However, with the passive case finding strategy, in a way the horse has already bolted because considerable TB transmission would've occurred before the diagnosis is made. These are individuals who would be spreading the bacilli. The second weak link in the chain, or key reason why diagnosis is problematic, is the inability to collect sputum samples. We already heard Sebastian talk about cavities, but not all TB patients produce sputum. And so getting a sample in about a third of patients is a problem. And this is because TB is either outside the lung, we call it exopthalmy TB, and second that somebody may not be producing sputum as such. And then thirdly, suboptimal diagnostic tools. And he alluded to smear microscopy where one looks down the microscope to identify the bug in, for example, a sputum sample, but that only detects about 50% of TB. We have DNA based tests, but these only pick up roughly about 80% of tb. And one can also grow the bug in the lab, but this takes many, many weeks and is also quite expensive.
James - So the problems are extensive and numerous. What are the most promising avenues to sorting this problem out? How do we get people diagnosed more quickly so they can get the treatment they need?
Keertan - Well, I think, again, going back to the three points I mentioned, the first thing is that we need to take diagnostics out into the community. And we've been doing research where we've developed a model where we take these DNA based diagnostic tests. These are portable diagnostic tests, and we take this in a mini mobile clinic. And, um, it's manned by two healthcare workers. And so we go out into the community to diagnose cases on site, we call that point of care diagnosis. We also do other research where we've developed or are developing, through the research teams at the University of Cape Town and the London School Sputum, Independent tests. And one of the tests we are working on is a protein based test. We discovered this using a mass spectroscopy approach. We can use, for example, a simple lateral flow assay using urine, for example, to rapidly screen for somebody in the community that has TB. The important point there is that one can rapidly identify individuals who might have TB and so target them for diagnosis and treatment. And we've also been doing work where we've developed a more sensitive tool called ERISA TB for a specific type of TB called exopthalmy tb. I had spoke about that early on. That's TB outside the lungs. And this is being scaled up through a spinoff company at the University of Cape Town. And we, for example, have managed to improve the diagnostic rate of extra pulmonary TB from about 30% using conventional tools to about 90%, which makes diagnosis much more accurate and efficient. So these are some of the examples of how one can make a more efficient and rapid diagnosis of TB.
James - Keertan, I'm afraid that's where we'll have to leave it. It sounds very promising. That's Keertan Dheda from the University of Cape Town.
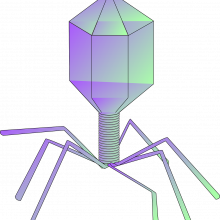
14:34 - Fighting TB with phage therapy
Fighting TB with phage therapy
Graham Hatfull, University of Pittsburgh
Treatment for TB is not trivial. Because the bacteria often adopt a dormant state in the body for at least some of the time, they are only periodically susceptible to antibiotics, which means that the drugs need to be given over an extended period of time; in some cases over more than 6 months. We also give a cocktail of drugs to gain rapid control over the infection, and reduce the risk of the patient developing an antibiotic-resistant infection. Nevertheless, we are seeing a significant increase in the prevalence of drug-resistant forms of TB, including those dubbed “extensively drug resistant”, which, unsurprisingly, can seriously limit therapeutic options. One glimmer of hope on the horizon is a technology that is actually older than antibiotics but enjoying a renaissance in recent years: phage therapy. This uses viruses that selectively attack bacteria but don’t touch human cells as a form of living antibiotic. Graham Hatfull, at the University of Pittsburgh, has been developing this line of treatment for bacteria that are in the same family (known as the mycobacteria) as human TB. He’s optimistic that phage therapy might give us a new weapon to fight TB too, as he explains to Chris Smith…
Graham - We were contacted first at the very end of 2017 by physicians treating a couple of patients in London that had infections with an organism called Mycobacterium abscessus. They had both had lung transplants and were taking immunosuppressive drugs in order to support the new lungs and got these disseminated infections. We were sent the strains to screen to see if we had any phages which would specifically infect those bacteria and after much screening we assembled a cocktail of three phages that were then administered intravenously with some evident clinical benefits, which we were pleased to see. Since then, we've had many, many inquiries as to whether we could do something similar for other patients because this is a fairly common scenario and I think that we've now treated or provided phages for treatment of about 40 patients in total.
Chris - And are they all doing well? Does it reliably work?
Graham - It's mixed. So we've published a report where we summarised the first 20 cases. Of those 20, 5 I can't really tell because other events happened. We can't really tell one way or the other. In 4 cases, patients did not obviously benefit, but in 11 we saw favourable clinical or microbiological outcomes, which we took as being promising, especially given that this was a cohort of patients who were extremely sick and literally have no other options.
Chris - And the infections that these patients had, were they what we would all call human TB or were they all members of the same family, but not specifically human TB organisms?
Graham - None of these cases are specifically human tuberculosis, but they do span a variety of closely related pathogens. Mycobacterium abscessus was the most common. There was one case of mycobacterium avian, and there was one case, which is the most closely related or relevant to tuberculosis, which was in an infection with actually a vaccine strain known as BCG.
Chris - And would you argue then that what you take away from the 40 people or so that you've managed so far with this approach, that that could now be applied to human TB?
Graham - For tuberculosis itself we're encouraged that there's very little variation amongst clinical isolates. And by doing extensive screening, we think we essentially have three to five phages that could be used as a cocktail and could be used essentially for all patients that have tuberculosis infection with mycobacterium tuberculosis.
Chris - And in practical terms then, how is this actually going to work and is it scalable?
Graham - The first decision that has to be made is what the root of administration is. For most of the cases that we've provided phages for treatment to date, they've all been by IV injection typically twice a day, but for extended periods of times - weeks or many months. So the idea with the intravenous administration is that the phages will get into the bloodstream, will find their way to the site of the infection and replicate on the bacteria killing the bacteria as they do.
Chris - So it's a long time, isn't it, to have to give these infusions if it does take months. Presumably this would be for a subset of patients, those that we can't manage with antibiotics.
Graham - So first of all, I think that at least early on, we would not want to rely on a long term therapeutic application with phages as a substitute for antibiotics. I would envisage them more likely being used in conjunction with antibiotics, which would at least have the prospects of shortening treatment and maybe helping to reduce the incidence of resistance to the antibiotics, which is clearly a major concern.
Chris - And what about resistance against the phage?
Graham - Great question. And we would anticipate resistance against the phages just as you see resistance against antibiotics. Our experience, however, with the treatment of these other types of infections that I mentioned, where we do have expertise is in treating some of these other infections and 11 patients, we've administered just a single phage. In those 11 cases, we've never seen any incidents of resistance. And in fact, if we look in the laboratory and we just challenge the bacteria with the phage and we ask, "How often does resistance occur?" it appears to occur at a very low frequency. So although resistance is a concern, our experiences suggest it may not be as big an impediment as we thought it might. However, we always are wary of it and try to adopt a strategy that if we have more than one phage that we can use in a cocktail, then that's going to greatly lower the occurrence of any resistance.
Chris - Do you have any feel for what happens with respect to dormant bacteria, which is ostensibly a big problem with TB; people catch it and may wall it off in their bodies in a viable but dormant state for an extended period of time. Can your phages get at those? Are they like the Heineken equivalent that refreshes parts that other beers can't reach? Can they get to these dormant bacteria and wipe them out?
Graham - Yeah, that's a great question. And of course, we don't know, but we would love to learn and find out about that. Our view would be that although we don't have good data on that, we are really optimistic about what the possibilities and the options are. Just imagine if you could take your bacteria phages and genetically modify them so that they are decorated with signalling molecules or small peptides that would target the phages very specifically to where the sites of those bacteria are. And so maybe we could actually design phages so they would specifically go after those guys that hide away in those parts that other beers cannot reach.
19:09 - Exploiting the chinks in TB's armour
Exploiting the chinks in TB's armour
Andres Floto, University of Cambridge
Another approach that scientists are trying to tackle TB is to explore ways to boost the immune system to better fight off the infection; there are also efforts underway to understand the molecular clockwork that makes TB tick in order to spot vulnerabilities that new antibiotics might be able to exploit. Chris Smith went to see Andres Floto, at the University of Cambridge, where he’s doing both…
Andres - It's a very attractive idea to see if we can find ways to stimulate the host immune system to either more effectively kill the bug at the very beginning or attack it in these walled off states. And so there's been a lot of interest to try and find ways in which we can target existing pathways within the immune system, stimulate them using drugs, and improve the ability of our body to kill these bugs. There are a number of ways in which you can do this and certainly in experimental systems and in pre-clinical models, these prove very effective.
Chris - How effective is very effective?
Andres - Okay, so our latest paper, which is currently under review, has identified a certain pattern recognition receptor. This is a receptor that identifies a certain compound produced by tuberculosis. And if you can stimulate that with drugs, you can improve killing in mouse models by three log orders. I'm trying to work out what that is...
Chris - A thousand times fewer bacteria will be left.
Andres - Yes, exactly. So certainly that is not proof that it's clinically effective, but the excitement is that there are a lot of drugs that are already in man for other indications, or indeed of being in clinical trials for other indications that could potentially be repurposed to solve this problem with tuberculosis.
Chris - Obviously the question stemming from that is, well why doesn't the immune system do that already?
Andres - It's likely that there are checks and balances that are occurring anyway as part of the way that the immune system is set up. But it's also very clear that tuberculosis has evolved to prevent these things from happening. And so we need to find ways in which we can supercharge pathways in order to overcome these inhibitory blocks and that's where you get successful killing. So we have a number of exciting candidates and none of these are yet in human trials, but watch this space.
Chris - Our experience with other types of microorganisms, particularly viruses like HIV, is that you hit them from multiple angles all at once and it has a much better chance of having a positive outcome. You stop the thing evolving or changing or becoming resistant and so on. Is it likely then that we'll couple what you are trying to do with additional antibiotic treatment?
Andres - So the two main goals of treatment of tuberculosis, one is to make treatment simpler and shorter. And the second one is to tackle these multidrug resistant organisms. And so in both cases you can envisage combining these what are called host directed therapies with either conventional or new antibiotics.
Chris - If you come along and stimulate people's immune systems really hard and then give them a load of these drugs on top, is that not going to make people feel pretty unwell and the compliance still may not be good?
Andres - So there's always a balancing act between stimulating the immune system in a way that's productive, that is to say, helps the body, and stimulating the immune system so you get an inflammatory response, which makes people feel terrible. We don't know yet because we haven't tested them in this context clinically, but there's an expectation that they should be well tolerated and if given at the right time with the right antibiotics.
Chris - Have we got any promising leads on the antibiotic front against TB? Because the World Health Organization, I know, are very worried particularly about multi-drug and and extremely extensively drug resistant TB at the moment.
Andres - So there's lots of efforts and the number of antibiotics in clinical trials has increased thanks to the efforts of the Gates Foundation and others. So it's very promising. What we've done in our lab is to think about ways in which we can rationally develop new antibiotics. If we can make generating new antibiotics easier by using structure guided design methods, then it should in theory allow us to just produce as many antibiotics as we need to multiple different targets within the bacteria. So that's very exciting. We've recently received funding from the Gates Foundation to understand what are the chemical rules to allow drugs to get into cells. And if we know that, then we can plan strategically how to design drugs that will ensure that they get into the bacteria.
Chris - I suppose this is a bit like looking at a clock and saying, how does the clock tick? If I can understand the ticking mechanism, I can understand where to throw a spanner to block it up most effectively.
Andres - That's right. The challenge with antibiotics is that the vast majority of them have been found in the environment as natural products. And actually if we try and screen chemicals against bacteria, we end up hitting the same target again and again and again. So as you say, if we understand the problem at a molecular level, we can then say, hold on a second, that gives us a roadmap to how to develop these antibiotics almost at will, which is very exciting.

23:14 - Are new TB treatments the real solution?
Are new TB treatments the real solution?
Anastasia Koch, Eh Woza & Sebastian Lucas
But despite these scientific steps forward, it’s worth bearing in mind that TB rates were declining in developed countries decades before antibiotics and even before scientists had actually even identified the TB bacterium itself. What caused that dramatic decline was not therefore modern medicine, but improvements in living conditions, a sentiment echoed by Anastasia Koch, co-founder of the non-profit Eh Woza in South Africa…
Anastasia - I don't believe that solving an infectious disease like TB is going to come only from a new biomedical intervention. And I think that's quite unusual for a scientist. I think HIV showed us, and then Covid showed us, that you can have really amazing biomedical interventions, like new antibiotics, new vaccines, but if people don't buy into it and you don't get people to trust you or trust the intervention, it's not going to have its intended impact. And I think in order to get people to trust you, you have to also address the social conditions in which the disease occurs. TB occurs within a bunch of other social conditions, and those are really important to address as well.
And pathologist Sebastian Lucas also emphasises the importance of good quality infection control as probably our best weapon against TB in the near term…
Sebastian - The secret to reducing tuberculosis impact globally is to have better surveillance so we can identify people who have tuberculosis, treat them properly, find who they have been with, in other words, contact tracing to see who they might have infected. And to do this very efficiently, we always need better drugs, and especially because of drug resistant tuberculosis, we would like to think an effective vaccine is on the horizon. Now, we've had BCG vaccination since 1921. This is derived from attenuated tuberculosis or microbacteria bogus, actually. But unfortunately, it is not terribly effective. It seems to have a little bit of an effect on whether a person who is infected actually develops disease, but it doesn't seem to have much effect on whether you actually catch TB in the first place. And so a lot of people have been looking for a long time for a better vaccine. And at the moment, I'm not aware that there are any good candidates around. And the final point relates to special populations such as those who have active HIV disease because they are particularly susceptible to a very severe form of tuberculosis and has a much higher fatality rate than in people who have not got an immunosuppressive condition like HIV.










Comments
Add a comment