Could a Siphon be used in orbit? Why do leaves change colour in Autumn? How is immunity passed from mother to baby through breastfeeding? Why do earthquakes happen away from plate boundaries? How do microwaves heat up food? We storm through your questions this week as well finding out how Twitter can be used to monitor moods around the world, how carbon dioxide can be converted back into a fuel, how biomarkers hidden inside ECG's can predict the risk of a repeat heart attack and how glowing bacteria can send secret messages! Plus, in Kitchen Science, we make flames without fire by making iron burn...
In this episode

01:36 - Probing the Formation of Mercury
Probing the Formation of Mercury
The Journal Science has published a special issue this week presenting the first 90 days of the MESSENGER probe's observations of the planet Mercury. The densest planet in the Solar System, Mercury is the only terrestial planet other than the Earth to have a strong magnetic field. Moreover, its surface is believed to offer a unique window on the early Solar System, because it has been geologically dead for at least 3.5 billion years, while the other planets have undergone subsequent volcanism, re-surfacing events and weathering. However, to date this enigmatic planet has proved difficult to study because it is so close to the Sun.
The densest planet in the Solar System, Mercury is the only terrestial planet other than the Earth to have a strong magnetic field. Moreover, its surface is believed to offer a unique window on the early Solar System, because it has been geologically dead for at least 3.5 billion years, while the other planets have undergone subsequent volcanism, re-surfacing events and weathering. However, to date this enigmatic planet has proved difficult to study because it is so close to the Sun.
It's still very early days in the analysis of data from MESSENGER, which only arrived in orbit around Mercury in March, and so what we're seeing at the moment are a number of strands of evidence, which still need to be interpreted, but which will feed into models of Mercury's formation in coming months. But those strands are quite tantilising. For example, the first measurements of Mercury's surface composition are quite a contrast to the compositions of the other terrestial planets -- high in sulphur and magnesium, but low in aluminium and calcium. That's a much closer match to the composition of comets: does it suggest that Mercury's surface may be littered with dust from comets? Particularly interesting is a high abundance of potassium, which is relatively volatile: its survival in Mercury's crust rules out any violent formation models in which Mercury would have been very hot at any point in the past.
Measurements of Mercury's magnetic field have shown a similar field pattern to the Earth's but displaced towards the north pole, so that the equator of Mercury's magnetic field is around 500 km north of its centre. MESSENGER has detected solar wind particles trapped within that magnetic field, and the surprise here was that the particles come in very short sharp bursts, typically lasting only a few seconds. This contrasts with solar wind particles in the Earth's magnetosphere, which become trapped for long periods in regions called the Van Allen belts, leading to a smooth and steady flow of particles. This suggests that Mercury's magnetic field is not strong enough to trap particles in the long term, and is much more exposed to the gustiness of the solar wind.
Finally, there have been some surprises among the features seen on Mercury's surface. Shallow irregular indentations seem to be common on the floors of craters. The mostly likely way that these might form is by the evaporation of sub-surface volatiles when new craters are formed, which might be expected as previously buried rock is exposed to the Sun's heat for the first time. This appears to be another indication that volatile materials are quite abundant on Mercury, apparently ruling out a hot and violent formation models.
The next step will be to feed these observations into more detailed models for Mercury's formation, and it will be interesting to see how these develop in the next few months.

06:08 - Designer plastics
Designer plastics
Plastics are used all over the modern world from toothbrushes to fighter jets, because they have a very wide range of physical properties. This is partly because there are lots of different plastics with different chemistries, but two plastics with the same chemistry but different structures can have very different properties. Plastics are polymers, they are made up of repeating chains, petrol, wax and polythene are all just different lengths of the same type of hydrocarbon chain. Plus the chains don't have to be straight, they can be branched, and the branches can be of different lengths, all of which affect the strength stiffness and other properties of the plastic.
This is a huge advantage, but it is also a problem, as normally plastics for a particular application are produced essentially by trial and error, testing lots of different conditions, amounts of catalyst etc, until they produce the right properties. This is particularly a problem if we are to replace conventional plastics with biodegradable or plant derived plastics.
Daniel Read and colleagues from Leeds and Durham universities may have made this process much more predictable. They have developed computer model which will take the conditions of a polythene reaction chamber and predict the degree of branching, and the structure of that branching.
They then feed this data into a second computer model which predicts the physical properties, from the molecular structure by predicting the amount the molecules can wriggle past each other, in the middle of the chains, and how easily the ends of the chains can move, and they have now got it to work very well for polythene.
This means that they could already predict the conditions you need to produce a polythene with particular properties, and potentially what happens if you blend different polythenes as you recycle them, and by changing a few numbers in their simulation they could adapt it for new systems like bio-polymers making their development much faster and more efficient.

09:42 - Monitoring Moods with Twitter
Monitoring Moods with Twitter
with Michael Macy, Cornell University
An analysis of over half a billion tweets worldwide has confirmed that regardless of country or culture, we’re all in a better mood in the morning. Speaking with Chris Smith, Michael Macy, at Cornell University, has analysed the messages posted on Twitter by 2.4 million people from 84 different countries to find out more...
Michael - We were looking to see how people's moods change over the course of the day, over the days of the week, and over the seasons of the year using Twitter which allows us to monitor people's expressions in real time at a global scale across diverse cultures, which is something that scientists have not been able to do in the past.
Chris - Talk us through the method. You presumably didn't read half a billion tweets individually?
Michael - That's right. It's all a process using computers. In fact, you wouldn't want to try this on your desktop computer - we used Cornell's Centre for Advanced Computing, a large cluster of powerful supercomputers, and what we did is basically for each user on a given hour in a given day, like let's say, Tuesdays at 9am to 10am, we would take all the messages that they wrote during that time period and throw them together kind of as a 'bag of words'. Then the computer sorts through that bag and identifies all the words that are positive words like 'awesome', 'fantastic', 'incredible', 'hilarious', and also finds all the negative words like 'unhappy', 'depressed', 'embarrassed', 'anxious', 'afraid', and so on.
Chris - How does it handle sarcasm? So if I said, "Brilliant! Just got fired." How would it cope with that?
Michael - It would get it wrong, and one of the advantages of having such a large number of messages is that the errors will tend to cancel out at very large numbers. So, you get a little noise with people using sarcasm, or for example saying, "Good morning" in the morning just as a ritual but not as an expression that they're actually feeling good. But when you have this large a set of observations, in this case over a half billion messages, then the errors tend to cancel out. The reason that we know we're seeing the errors cancel out is that we get a very robust pattern - it holds up. We see the same thing each day of the week, and we see the same thing across very diverse cultures.
Chris - And since you say you saw these patterns, what patterns did you see? What did the analysis reveal?
Michael - Well basically, we found that people are happiest in the mornings and then it's sort of all downhill from there! But then, in the evening, there's a rebound in positive feeling, right on up to bedtime with a second peak in the late evening. And we see this pattern every day of the week so it's not just work that's doing it, but it certainly fits with what we think about work -something that makes us tired, it gets us stressed out, the frustration of commuting, and so on. We actually see the same pattern on the weekend, so these two peaks with a trough in the late afternoon seems to be something that's kind of built-in to human body rhythms and it's independent of the day of the week or whether you're at work or not. On the other hand, people are also happier on the weekend which does suggests that there's something going on with work, but the peak on the weekend is a little bit later. It's about one and a half to 2 hours later than it is on a weekday which suggests that people are sleeping in, whereas on the workday, they're getting woken up by an alarm clock. On the weekend, they're perhaps sleeping until their body is ready to wake up, and so it could be that this weekend effect, the elevated mood on the weekend, is actually a response to having sleep that is not interrupted artificially by an alarm clock.
Chris - It also tells me that the people you analysed in your study obviously didn't have kids because they wouldn't have been afforded the luxury of a 2-hour lie-in! What about one other quite important thing which I guess you may have been able to test this with this sort of data set - seasonal affective disorder. The whole idea that as we shift through the seasons, people at very high latitudes are being exposed, not just to an absolute reduction in day length but to a changing day length. Do you see this mirrored in the moods indicated by your analysis?
Michael - Yes, we do. But it's actually not the absolute amount of daylight that you're getting or how long the day is in absolute terms. It is the change from what you're used to. So, what's happening is that as the days are getting longer, moods are improving and as the days are getting shorter, then moods are not so good, and it's the change in the daylight, not the absolute length of the day that seems to be associated with changes in mood.
Chris - So what's the bottom line with this study? Why is this important? What have you flushed out from doing this that we didn't know previously from other sociological experiments on small groups?
Michael - Because we had such a large set of observations from all over the world, we're able to have more confidence that we're nailing down when in fact it is that people are in the best mood. We also had some surprises in the results - finding the same pattern on the weekend and finding the same pattern across very diverse cultures was something of a surprise. You would think a country that's collectivist for example, or has a different religion from the US, UK, or Australia, you would think that perhaps they would have different patterns of when people are in a good or bad mood. But in fact, we found very similar patterns all across the globe - whether it's India, or Africa, or the UK or the US, Australia - very similar patterns.
Chris - What are the implications of that?
Michael - Well one of the things we'll want to do is just to break it down a little bit into more detail of the different groups within this large population in different ways. So for example, one of the things that we looked at that we might want to investigate further are different chronotypes. A chronotype is a person who's active at a particular time of day such as a night owl or a morning person. Another one of those surprises was that we found out that night owls are different from everybody else in a sort of peculiar way. Everybody else has their second peak when their moods rebound in the evening, and you would think that certainly night owls will too, but in fact we found the opposite. The night owls just have that peak in the morning like everybody else, but they don't have that rebound in the evening. They're basically the group that's the least happy in the evening and yet they're the group that's the most active. So that was a bit of a surprise and we'll pursue in more detail those kinds of breakdowns of who in the population is reacting in what way.
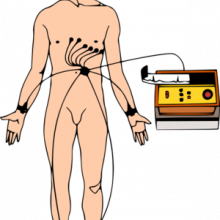
16:56 - Getting to the heart of coronary risk prediction
Getting to the heart of coronary risk prediction
An analysis of electrical heart tracings from thousands of patients has 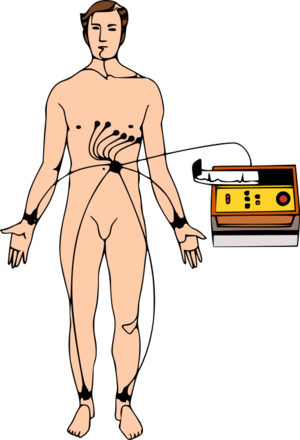 revealed a new way to spot those at risk of repeat heart attacks.
revealed a new way to spot those at risk of repeat heart attacks.
University of Michigan scientist Zeeshan Syed and his colleagues, writing in Science Translational Medicine, used a computer to marry up clinical outcomes with the features present in long-term electrocardiogram (ECG) recordings of over 4500 people who had undergone a recent heart attack.
They were able to define three characteristic "biomarkers" that could be used to predict an individual patient's risk of further heart problems. These they have called Morphological variability (MV), which looks at how the shape of the ECG tracing changes over time, the Symbolic Mismatch (SM), which compares an individual patient's ECG trace with those of other patients with a similar clinical history, and the Heart Rate Motif (HRM), which looks at how often "risky" patterns of heart rate activity crop up in an individual's recordings.
Incorporating these assessments into the existing protocols used to calculate a patient's risk could, they say, enable doctors to better identify those patients most in need of close monitoring or more intensive drug therapies; on the flip side, it could also potentially spare patients who are actually at a lower risk than first thought from having more rigorous interventions that they don't require. Moreover, as the scientists point out in their paper, "these biomarkers can be extracted from data that are already routinely captured from patients with acute coronary syndrome and will allow for more accurate risk stratification and potentially better patient treatment."

20:36 - Space Stations, The Dead Sea Scrolls and Glowing Bacteria...
Space Stations, The Dead Sea Scrolls and Glowing Bacteria...
with Anu Ohja, National Space Centre; Geza Vermes, University of Oxford; Rich Masel, Dioxide Materials; Michel Lucas, Harvard School of Public Health; David Walts, Tufts University;
Chinese Satellite Launched
Meera - China has launched its first space station, set to orbit 220 miles above the Earth.
The unmanned Tiangong 1 space laboratory, meaning 'Heavenly Place' was launched from the Gobi Desert on the 29th September carried up by the rocket, Long March and will remain in orbit for 2 years...
Anu Ohja from the National space Centre in Leicester, on the aims of the mission...
Anu - In a couple of month's time in November, there'll be the mission called Shenzhou VII, practicing rendezvousing and then docking with Tiangong-1. Now that's going to be unmanned and the plan is for that to stay up there for 12 days. What will then follow over the next year are two more missions. One of which will be manned, but the current plan is to have Tiangong-1 altogether in orbit for about two years with Tiangong-2 following as a successor in 2014.
---
Dead Sea Scrolls Online
Meera - High Resolution images of the Dead Sea Scrolls are now available online.
Five major scrolls from these ancient manuscripts, originally written on parchment and papyrus, have now been digitised by Google in collaboration with the Israel Museum, enabling billions of people to access these biblical texts in their original form, as Geza Vermes, Emeritus Professor of Jewish Studies at the University of Oxford explains...
Geza - One of the most important contributions here is, that people can see the actual scrolls as they are, the colour of the leather, shape, darkness, and spots of ink on the manuscript so that they can actually come as close to the documents as those who can hold them in their hands.
---
Converting CO2 emissions into fuel
Meera - Carbon Dioxide emissions could soon be converted back into fuel, reducing levels of the greenhouse gas released into our atmosphere.
Using a process dubbed artificial photosynthesis, Rich Masel and colleagues at the company Dioxide Materials, have used solar energy to convert carbon dioxide into carbon monoxide and water, which in turn combines with hydrogen to produce synthetic gas now used by many oil companies to produce fuel...
Rich - This is a viable route in the long term to make the fuel for your car, or your truck or your airplane that doesn't compete with the food supply, and it's completely renewable and recyclable. So it changes CO2 from something we dump or have to bury into something that we recycle and reuse, and it's a renewable resource.
---
Hidden Messages with a Bacterial Glow
Meera - Glowing bacteria could soon be used to encode secret messages.
 Working with E. coli and fluorescent proteins, David Waltz from Tufts University used combinations of bacteria to encode messages onto sheets of paper made of nitrocellulose. Potential uses for the process are product authentication, prevention of counterfeiting and even biological bar coding, with people wanting to decide for the message, needing to know the right nutrient and light needed to read the bacterial glow.
Working with E. coli and fluorescent proteins, David Waltz from Tufts University used combinations of bacteria to encode messages onto sheets of paper made of nitrocellulose. Potential uses for the process are product authentication, prevention of counterfeiting and even biological bar coding, with people wanting to decide for the message, needing to know the right nutrient and light needed to read the bacterial glow.
David - It's a combination of two colours, corresponds to either a letter, a number or a symbol, and we array those in a particular order and bacteria do not glow unless they're exposed to a particular nutrient. And so, when those bacteria grow in the proper medium, they develop the colours that they're designed to present and the message, kind of like invisible ink then appears magically.
---
The Ig Nobel Awards
And finally, the 21st annual
Ig Nobel awardstook place at Harvard University this week honouring scientists for research that makes people laugh and then makes them think.
This year's prizes honoured work on why discus throwers get dizzy when hammer throwers don't, the use of wasabi to wake people up in an emergency, as well as our decision-making ability when needing to urinate.
Marc Abrahams is in charge of the ceremony...
Mark - The point of them in a way is just to show people things that are so unexpected and entrancing. They are going to make you want to know more. Among the winners were the Mayor of Vilnius, Lithuania who won the Peace Prize for demonstrating a new way to discourage people from parking their cars in bicycle lanes. He runs over their cars with an armoured tank.
The prizes themselves are presented by real Nobel Prize winners and continue to grow in popularity each year.
Can you cut down energy consumption by altering the power supply?
Chris:: Okay, so just to clarify, you put these things into your electrical supply and in your case, you're saying that you're making geysers or in other words, hot water immersion heater sort of things, and they're supposed to cut down the amount of energy you're consuming in the home. Dave, what do you think?Dave:: There's a couple of ways of doing this - one of them is called 'Power Factor Correction'. This is, if you've got very large motors or sometimes very large electronic loads, you can draw current not at the same time as voltage. So if you imagine alternating current - the current voltage is going up and down, up and down - and if you attach it to a light bulb then it will draw a current at the same time as the voltage, and that's fine - that's actually what the power company wants you to do. But sometimes, if you have these strange loads, they can actually draw lots and lots of current when the voltage is very small, and draw very little when the voltage is very high. That means that because your meter measures a total amount of current (which is flow multiplied by the time) you're apparently using far more energy than you actually are, so you get charged more than you should be, so it can reduce what you're being charged. It also drives the power companies up the wall because it causes havoc with all their systems!Chris:: But moreover, it means that they're doing less for their shareholders than they were before.Dave:: Yeah, I mean, despite the fact that this way you get more power, then they're happier. But that's only a really big issue if you've got lots of big three-phase motors, big motors, or all possibly a very large amount of electronics. But I doubt that's a big issue with houses.Chris:: So Roy's immersion heaters are probably not going to benefit from having one of these things then?Dave:: Certainly not from that. The other way then that some of these things can work is by essentially just reducing the voltage. So it's basically like putting a variable transformer into your system. So, whatever the voltage is coming, it will always put out 220 volts. That means your geysers will actually be less powerful so you'll be able to use less power, but that means you also get less heat.Chris:: You're just going to use it for longer, aren't you?Dave:: But there are some kinds of lighting, especially fluorescent lighting, that are more efficient that way and things like transformers are more efficient that way. So, it'll produce a big reduction in the amount of power you use, but you might get less out of it as well.
Can planets form from asteroids?
Dominic - This is one of those really good questions where I have to say we don't completely understand the answer. The way that we think planets formed in our solar system and elsewhere is by solid particles of grit-like material which came together and formed into small asteroids, and then those asteroids gravitationally attract one another and come together to form large masses that turned into planets. Now for some reason, we have this gap in the middle of our solar system between Mars and Jupiter, and it's a big gap in which there are no planets, but there are lots of these small asteroids. So, given that no planet has formed in the last 5 billion years since the solar system formed, we can probably say there is some good reason why a planet has not formed there, and probably the reason is that those asteroids are too close to Jupiter, and Jupiter is stirring up the orbit of those asteroids and stopping them from gravitationally attracting to one another and coming close enough together to form together into a planet in that gap between Mars and Jupiter.Chris - There was a terrific paper in Science about two years ago by David Minton who was I think in Arizona and he did a mathematical model, another computer model of the Kirkwood Gaps in the asteroid belt. There are all these holes in the asteroid belt which can and can't be accounted for and they worked out where Jupiter had come from and where it had gone to, because it didn't always end up in the same position it's in now. It's migrated a bit during its evolution of the solar system. And this has created, when they put it into their model, what they dubbed 'gravitational resonances'. So it lined up with all the other planets in the right sort of place, it shook the asteroid belt a bit and dislodged some of these other bodies from the asteroid belt, kicking them onto earthbound orbits actually and probably accounted for the late heavy bombardment that rained in on the planet 3.9 billion years ago. But their point was that the gravitational tug there is so unpredictable and so intense that it actually stopped all these things accumulating, and accreting into a planet, or even if they did at one time, they fell apart again.Dominic - Yes, that's absolutely right. The Kirkwood Gaps are really the forensic evidence that Jupiter is the problem why no planet has formed there, because the Kirkwood gaps are lined up with places in the asteroid belt where you would be orbiting twice as fast as Jupiter or perhaps 1.5 times as fast as Jupiter. And for some reason, there were no asteroids there and that suggests that there is some reason why you can't be in orbit at that point.

Why do earthquakes occur far away from plate boundries?
Dave Ansell considered this question...
Dave - Yes, Washington DC is nowhere near a plate boundary. There's a variety of things and, basically, anything which can apply large forces to the plates can cause earthquakes. You can also get earthquakes a long way away from plate boundaries, actually still associated with them.
So, in India, where you're creating the Himalayas and you've got a huge amount of compression, you can still get earthquakes hundreds of miles away from that plate boundary, just because the compression and the crushing there is so immense.
You can also get earthquakes when people build big reservoirs: so you put a load of water on the ground. That essentially loads it, so the comfortable place where the ground wants to be is a bit lower, so the only way it can move there - if it's not squidgy like clay - is by cracking, and so you get small earthquakes like that.
You can get earthquakes as ice melts - we're still getting earthquakes in Norway and actually, most of the earthquakes in Britain are probably associated with all the ice melting after the last ice age. In the north end of Britain, and in Norway, the ground is rising slowly actually a few centimetres a year, especially in Norway, and that eventually builds up some pressures and stresses in some places until it breaks.
I think the one thing I vaguely heard about the Washington earthquake is that it could also be sediment building up off the edge of the continent, building up on the ground that's pushing down and, eventually, that could build up enough to create an earthquake...

Do babies acquire immunity from breastmilk?
In many young animals the intestine is set up to collect antibodies present in maternal milk and to pass it into the bloodstream to provide so-called "passive immunity" while the infant animal's own immune system becomes established. But, we don't believe human babies do this to a great extent*. This is not to say that the antibodies in breast milk are useless though. Far from it; the bulk of breast milk antibodies are what we call IgA; these are secreted antibodies and specialised for defending body surfaces. After the infant consumes them they adhere to the gut wall where they form a strong line of defence against a range of pathogens.
The major way that antibodies get into a young baby are via the mother's placenta, because once a baby goes beyond about 28 or 29 weeks of gestation, the placenta turns on special receptors called FC receptors which grab from the mother's bloodstream antibodies of a class called IgG, and they put them into the baby's bloodstream and that way, when the baby comes out, it's 'passively protected'. In other words, it's got antibodies recognising all the things that its mum is normally being exposed to in the environment in which she lives, so it's a good way of protecting the baby for its first weeks of life because those antibodies circulate for up to six months; after that time the baby should be able to stand on its own two immunological feet.
Unfortunately, although antibodies cannot cross the newborn gut wall, this is not true of everything, and some viruses, including HIV, can infect an infant via this route. In fact, the risk of a baby catching HIV from a mother if it's born vaginally and at term is actually quite low - about 5%; but if the mother breastfeeds the baby the risk can go up quite significantly - to over 20% - so not breastfeeding in those circumstances can actually spare quite a lot of infections.
[Note added later: I'm grateful to Michael Lindenmaier for writing to me about this question and also sending me this reference, which is pertinent to the subject].
"Differing from humans, IgG from breast milk in many animal species (rodents, bovines, cats, ferrets, etc.) are transported across the intestinal epithelium into the neonatal circulation. This transport is located at the duodenal and jejunal level where enterocytes express a surface membrane receptor able to bind Fc of IgG and to facilitate transcytosis of these immunoglobulins. Fcγ-R, which is very similar to the placenta receptor responsible for active transplacental transfer of IgG in humans, binds IgG but not other isotypes. Maternal milk antibodies represent an important part of circulating IgG in these animals, as they are involved in the negative feedback of endogenous IgG synthesis. This phenomenon stops abruptly as soon as weaning takes place. Neonatal calves that have a defect in such transfer of maternal immunoglobulins are at high risk of systemic infectious diseases.In humans, in whom gut closure occurs precociously, breast milk antibodies do not enter neonatal/infant circulation. A large part of immunoglobulins excreted in milk are IgA that protect mainly against enteric infections. The specificity of maternal milk IgA is driven by an entero-mammary cell circulation. Human milk also contains anti-idiotypic antibodies capable of enhancing infant antibody response. Maternal milk antibodies coat infant mucosal surfaces and some have a clear protective role."
From: Transfer of antibody via mother's milk, Philippe Van de Perre (2003), Vaccine 21: 3374-3376
*Note that, in an interview with Caltech scientist Pamela Bjorkman in 2008, we heard how some IgA is picked up and moved into circulation by intestinal cells.

38:33 - Exploring Lake Ellsworth
Exploring Lake Ellsworth
with Chris Hill and Martin Siegert, University of Edinburgh
Martin - Lake Ellsworth is at the bottom of the west Antarctic ice sheet. It is underneath about three kilometres of ice. The lake is roughly ten kilometres long, two or three kilometres wide and the water depth of it is about 150 metres. So it's like a rather large Scottish loch under the ice of west Antarctica.
Richard - Why is it water?
Martin - Very simple - the background geothermal heating is all you really need to get the ice temperature to be at the pressure melting point beneath about three kilometres.
Richard - So that's heat from the rock?
Martin - Yes, it's not unusual geothermal heating, it's just background levels of geothermal heat that you get everywhere on the planet and that's sufficient to warm the ice to the level that it will melt, and it's expected in Antarctica. We know of about 400 subglacial lakes that are located in various parts of Antarctica. Lake Ellsworth is the one that we're investigating in the next few years.
Richard - Why is it so interesting?
 Martin - Well there's a number of things that we want to understand about subglacial lakes. It's been 15 years since a breakthrough paper highlighted the scientific interest in subglacial lakes being a place where life might exist - unusual microbes in a very extreme environment - and we're interested in understanding how life can survive in these places, what life exists in these places, whether it's thriving or whether it's on the edge of extinction, and also the sediments on the floors of subglacial lakes may give us very important information about past climate change and ice sheet change in Antarctica.
Martin - Well there's a number of things that we want to understand about subglacial lakes. It's been 15 years since a breakthrough paper highlighted the scientific interest in subglacial lakes being a place where life might exist - unusual microbes in a very extreme environment - and we're interested in understanding how life can survive in these places, what life exists in these places, whether it's thriving or whether it's on the edge of extinction, and also the sediments on the floors of subglacial lakes may give us very important information about past climate change and ice sheet change in Antarctica.
Richard - So what are you planning to do?
Martin - Our plan is to access and sample the lake water and get samples of the sediment as well, and to do that is rather difficult because we have to get all the way through that three kilometres of ice in a very clean way without disturbing the lake unduly and getting those samples back to the surface where they can be treated and analysed in laboratories.
Richard - Because there might be life there, you don't want to contaminate it, and you want to ensure that if you find traces of it, it's from the lake and not something you've put in there?
Martin - Absolutely right. But science demands that we do things cleanly as well. We're very likely to be encountering very low concentrations of microbes and chemicals that we want to measure, and unless we keep the experiment really clean all we will do is measure the things we take down with us.
Richard - Now, Chris, it's your responsibility to make sure this happens?
Chris H. - That's right.
Richard - When we talk about drilling down, you're actually using hot water to drill?
Chris H. - That's correct. Traditional drilling techniques would have used some sort of drill corer but that takes a long time and hot water drilling is actually the cleanest, quickest and most efficient way of accessing the lake.
Richard - So what have we got here? I mean these really are big paddling pools. There are two of them set up at the moment and they're full of water and I think it's got quite a nice sound when you tap the side. These are the pools that you have hot water in to drill down?
 Chris H. - Kind of. What we need in order to undertake hot water drilling is a huge amount of water and of course there's loads of water in Antarctica - The problem is it's all solid! So the very first thing we need to do is to get together a large amount of snow in one place and start to melt it and that's what these pools are for - to melt some 90,000 litres of water to prime the drilling system to start drilling. Once we are drilling, there's no problem because as we're drilling down to the lake we are generating lots of melted snow which as we go which we can recirculate.
Chris H. - Kind of. What we need in order to undertake hot water drilling is a huge amount of water and of course there's loads of water in Antarctica - The problem is it's all solid! So the very first thing we need to do is to get together a large amount of snow in one place and start to melt it and that's what these pools are for - to melt some 90,000 litres of water to prime the drilling system to start drilling. Once we are drilling, there's no problem because as we're drilling down to the lake we are generating lots of melted snow which as we go which we can recirculate.
Richard - The drill's really a jet of water at the end of these giant hose reels which you've got trailing across the field here?
Chris H. - In simple terms, the system is really straightforward. We take this melted snow on the surface and the first thing we do is filter it. Then the next thing we do is heat it through a very clean boiler system up to approximately 97 degrees Celsius. We then run it through a bank of high pressure pumps and then, basically, push this water through a very long hose, a 3.4 kilometre hose, which has a jet nozzle on the end which just allows us to melt through the ice at an incredibly fast rate.
Richard - Martin, you've got your hole, what do you then do?
Martin - We have two items that we'll send down. One is a probe which has sample chambers on it and that will be lowered down the water column taking measurements. As it does so, it will scrape the sediments on the floor of the lake and it will take samples as it comes up and we will take that back to the surface and bring them back to the laboratories. The next thing we will do is put a coring device in which we can take a three-metre core of the sediments from the floor of the subglacial lake and transport it back to the laboratories.
Richard - It sounds to me almost like a space mission!
Martin - It's also another analogy to space science that the experiment that we conduct in Lake Ellsworth at some stage in the future may well be done on Europa, which is an ice covered moon of Jupiter with an ocean underneath it. People speculate that there may be life on Europa. To test that hypothesis will require more or less the experiment that we're going to do in Antarctica. It's maybe 20, 30, 50 years away, but we're very keen in Lake Ellsworth to make sure that we're documenting everything that we do so that in some stage in the future, if people want to learn how we did this experiment, it will be available to them.

Burning Iron - Igniting wire wool

49:44 - Why microwaved soup heats up faster at the edge?
Why microwaved soup heats up faster at the edge?
Dave - It might be something to do with a slight misconception of how microwaves cook...
People often say they cook from the inside out - they don't, strictly. Actually, microwaves are form of electromagnetic radiation, a form of light, and they're absorbed within the outside inch and a half or so of the food.
So, right in the centre, it's kind of insulated from the microwave by all the other food around it. So the soup in the middle is going to be getting less heating than the soup on the outside. Also, the soup on the outside is thinner so it's going to get heated from all directions. The soup on the outside is essentially getting more microwaves than the soup in the middle, so it gets hotter.
Chris - Thank you, Dave. And what can I do to remedy the problem so that my soup is more evenly heated in the future?
Dave - Take it out occasionally and give it a mix is your best bet.
Chris - I'll remember that!

50:46 - Would swimming from a submarine cause the bends?
Would swimming from a submarine cause the bends?
Chris - This is a good question, one I've actually pondered on myself which is why I guess it's come my way.
The answer is, probably not. Now the reason for this actually is that, when you're on a submarine, the air that you're breathing in the submarine is not under pressure or at least not under demonstrably higher pressure than ambient pressure - ie surface pressure - because the submarine can be thought of as an incompressible tin can underwater; so although the water is pushing in very hard on the submarine, the air inside is not feeling any extra pressure when the submarine goes down to the bottom of the ocean.
Therefore, you're breathing air as though you're breathing at the surface, and that means, if you escape from the submarine - say you went up one of the torpedo tubes or something in a submarine that was stuck underwater - then although you would immediately be exposed to extremely high pressure, depending on how deep the submarine was, then pressed in on you would be the surrounding water pressure and it would be subjecting the air already in you to that higher pressure; this means that the air would dissolve more in your blood, including the nitrogen that was in your lungs at that time, but there won't be very much of it. And, as you went up to the surface, you would find that nitrogen coming back out of solution and back into your lungs, and back into your blood, but again, there would only be a very small amount of it.
If you were a scuba diver, on the other hand, you would have a problem. What happens there is that scuba divers go down to the bottom of the ocean - say, 40, 50 metres underwater - and they're breathing compressed gas (in order to inflate the diver's lungs, to compete with the surrounding ambient pressure underwater, you've got to deliver the gas at higher pressure than the surrounding water) - therefore, you're breathing gas which becomes increasingly dense and increasingly high pressure the deeper you go; therefore more of the oxygen is going to dissolve in the bloodstream the lower down you go, but also the nitrogen is going to be forced into solution. Nitrogen is not very soluble, so then when you come back up again, because you have a body which has a lot of dissolved gas in it, as the pressure comes off, that nitrogen comes back out of solution and it forms bubbles in the tissue, and you get the bends.
If you're not breathing compressed gas, this won't happen. Whales don't get the bends or at least not often unless they resurface incredibly rapidly. We've made a very nice video of this. If you look at Naked Scientists Scrapbook on YouTube, you can actually see the footage we did recently - it's 'Why Don't Whales Get The Bends But Divers Do?' and it goes through the physiology of this.
Dave - So the problem with the divers is that their breathing air compressed for a very, very long time, whereas if you escape from a submarine there are only a couple of lungfuls at most, and there's not enough time for it dissolve?
Chris - Exactly. You're breathing air which is under pressure so it's pushing a lot more nitrogen to dissolve and nitrogen doesn't like dissolving. It's very insoluble. You also can't metabolise it, so even if it's in a tissue somewhere, it won't get used up. So you're increasing your total body load of nitrogen because you're breathing compressed gas, and the longer you spend underwater, the higher the burden of nitrogen in your body. As you go back to the surface, the pressure on you comes off and that pressure that was keeping the nitrogen dissolved is now gone so the nitrogen comes back out of solution and turns into bubbles again.
Dave - But I guess if you escape from a submarine, at some point, you're going to have to take a breath of compressed air, otherwise you're going to get flattened when you get out under high pressure so you come out through sort of an airlock or something so you'll be breathing compressed air for a bit.
Chris - That's true. As the, say, torpedo tube floods, it's going to compress the air in there and that will in effect subject you to compressed air. If you don't breathe it, then what I say, stands. If you do breathe it, you would have a little bit more nitrogen in your body but assuming you did it only for a very short time, I don't think it would make very much difference really.
Why do mornings get shorter and afternoons longer during the Winter solstice?
Dominic - That's a really tricky question. What it comes down to is how we define the length of a 24-hour day. The traditional way that you would do that is you would look at the sky and you would look at when the sun is highest in the sky and you will call that noon - the middle of the day - and then the next day you would go out and you look up when the sun was highest and that would be the next noon. The other way that you can define length of the day is to have an atomic clock which is measuring the progress of the laws of physics. It's giving you a very precise packet of time of a fixed length. Now it turns out, those two definitions of length of day are not exactly the same. The length of time it takes for the sun to come around from one noon to the next varies over the course of the year, and the reason is because that period is not just the time it takes for the Earth to rotate on its axis, but also the time it takes for the Earth to catch up with the fact the sun has moved a little way across the sky. The way in which the sun moves across the sky changes at different times of year because, for example, the Earth's orbit is elliptical and so the Earth is moving at different speeds at different times of year.Chris - I did hear one person put this as a bit like slipping a gear because you've got an ellipse, not a circle. As the sun goes along the flat side of each ellipse, it's got to sort of slip a gear a bit. The Earth has got to slip some gears as it turns in order to make it work, if you see what I mean. It's not just a straightforward circle.Dominic - That's not a bad way of thinking about it. Now what that means is that noon doesn't always happen at exactly 12 o'clock on every day of the year. For example now, at the beginning of October, noon is happening about 10 minutes before 12 o'clock via a watch and it's getting earlier. In a couple of week's time, it will be at quarter to noon, and then it will turn around and it will start getting later as the durations between noons get longer in December towards the winter solstice. So what's happening at the winter solstice is that sunset turns around in the middle of December, but sunrise turns around about a week later because the solar day - the time between noons - is drifting through the mean day that we call civil time.Dave - So the length of day is changing and the time of noon is changing, so the two add up to this effect?Dominic - Yes, that's exactly right.

Do other Solar Systems have asteroid belts?
It's really too early to say.
We've seen planets around several hundred actually, other stars in the last 15 years or so, but the mass of the asteroid belt is quite small.
I don't think that you really have the sensitivity to test something that's that small and which is that spread out across the place that you may need a big concentrated planet which is having a gravitational influence on its host star.

58:39 - Why do leaves change colour in Autumn?
Why do leaves change colour in Autumn?
The answer to this is that leaves look green because they contain the pigment chlorophyll, which is what they need to do photosynthesis, the process of grabbing energy that's in sunlight and driving a chemical reaction with it in order to turn carbon dioxide and water into glucose - C6H12O6; that's what photosynthesis is.
But leaves also contain other chemicals, including antioxidant chemicals and one of the chemicals they contain is a family of chemicals called carotenoids which, as the name suggests, are "orangey" or yellow.
As we get towards autumn, because the leaves know that they're senescing - they're getting old and they're going to be lost soon - they reduce their rate of chlorophyll production, and the tree removes the chlorophyll break-down products. That means that there's less of the green pigment in the leaf and therefore the yellow colour that the green was previously hiding is disclosed.
This is why the leaves appear to change colour, because the leaves are running out of chlorophyll.
Some leaves also turn a red colour though, don't they, which is nice. Not all leaves, but some species and that's because actually, they make another class of chemicals called anthocyanins. These are the same things you find in beetroot. They're a dark deep red colour and they're also a family of antioxidants.
The plants put those into the leaves to sustain and support them and prevent stress in the leaves as they go towards winter; that means the plant has longer to scavenge back from the leaf the things that it wants to rescue back into the plant before it dumps the leaf - because once you lose the leaf, you're losing tissue, you're losing salts, and chemicals, and potentially, other good for you things.
So, by protecting the leaf with these other anthocyanin molecules for a little bit longer, you hang on to your leaves for slightly longer than you otherwise would. Again, loss of the green chlorophyll molecules reveals these other coloured pigments hiding in the background...

Would a siphon work in space?
No it wouldn't, because you need gravity to make that siphon work. If you imagine the fluid at different points along the length of that siphon, at some points the siphon will be going uphill, and at other points it will be going downhill, and there'll be gravitational force on the fluid inside your siphon. And where the siphon is going uphill, the force is pulling the fluid backwards along the pipe. When it's going downhill, that force is pulling the fluid forwards along the pipe and what you need is more bits of length of your siphon going downhill with those forward forces than you have going uphill. And that means that because of the water pressure inside that siphon, the places where the force is going downhill is dragging fluid along the whole siphon from the beginning to the end. On the International space station, you have no gravitational force so there'd be nothing to drive the fluid along the siphon.
How does Rain-X work to help you see better through your windscreen in the rain?
The first thing it's doing is covering your windscreen with something called a fluorosilane. It's basically silicon-oxygen-silicon-oxygen, with fluorines on the side. It's incredibly hydrophobic so the water droplets, instead of forming kind of flattish lenses, form very, very circular things. I think what's going on is they're much, much stronger lenses. So they bring any light to a focus, very, very close to them and then the light spreads out, is evened out all over everywhere. So instead of using a really bright light, you see a kind of a much, much dimmer light and you hardly see them at all. And then probably, when light comes very close to you, something more complicated happens and you get the bright lens.

63:27 - Does a pregnant mother have to breathe more?
Does a pregnant mother have to breathe more?
We Posed this question to Dr, Gerald Hackett from The Rosie Maternity Hospital in Cambridge...
Gerald - Good afternoon. I'm Gerry Hackett. I'm a Consultant Obstetrician and Gynaecologist who work at the Rosie. Amongst many other things, I'm a Foetal Medicine Specialist and you've kindly asked me a question about pregnant mums and do they have to breathe more. Certainly, during pregnancy, a mother and a baby need more oxygen. Actually, many people think that the oxygen is just for the baby, but actually, at least half the extra oxygen the mother is taking in is for the placenta. Certainly, also during pregnancy, the mother's basic metabolism just gets higher. During pregnancy, mothers do breathe more. They in fact breathe more deeply. They don't breathe more often, more frequently, just more deeply. And actually, strangely enough, despite taking up extra oxygen in her blood, mothers in pregnancy often feel breathless and that's one of the most common symptoms of pregnancy, is breathlessness strangely enough. So during pregnancy, mothers will feel more breathless that even though they're taking up more oxygen, and letting out more carbon dioxide, they do this in part by movement of the diaphragm. Of course, as the pregnancy gets larger, you'd expect there to be less movement in the diaphragm. But in fact, there's just as much as always and so that tidal volume, the amount of air that a mother will breathe in and breathe out is much the same in pregnancy as outside of pregnancy. The airflow is much the same. The extra oxygen passes through the placenta, is taken up by the placenta, but also by the baby and it transfers quickly over to the baby's side because the haemoglobin - that is the red cells actually in the baby's circulation- picks up oxygen much more easily than our own haemoglobin would do. It binds that oxygen and it releases it into the baby's circulation. Diana - Pregnant women don't breathe more often, but they do breathe more deeply. And surprisingly, the movement their diaphragm can make isn't impeded by the growing baby. And on the forum, CliffordK said their adding 50 pounds to anybody or make it take more energy to walk across the room and oxygen is required to release that energy.










Comments
Add a comment