We often hear about amazing new medical developments which could improve disease treatment. But what about the ethical considerations involved in deciding how to use these advances? Hannah Critchlow and Ginny Smith discuss how we decide which drugs we can afford and what the limits are on designer babies. Plus how DJ's help get you in the groove, the risk of dengue fever at the World Cup, and how you can win the Ã?,£10 million Longitude prize!
In this episode

01:02 - Get your feet to dance to the beat
Get your feet to dance to the beat
with Dr Mark Elliott, Birmingham University
 Ever been on the dance floor, getting your groove on, enjoying a particular tune, and then the DJ flips the song over, and it completely messes up your dancing moves? I have! Then there's been other times when I haven't even noticed the tune changing
Ever been on the dance floor, getting your groove on, enjoying a particular tune, and then the DJ flips the song over, and it completely messes up your dancing moves? I have! Then there's been other times when I haven't even noticed the tune changing
But what's the science behind this DJ skill? Well, Mark Elliott, Research Fellow at Birmingham University thinks he's found the answer.
Hannah - Hello. So, what makes a good DJ?
Mark - So, a good DJ basically needs to mix two songs together seamlessly. So basically, it will sound like this...
(sound)
Mark - So, they've got to take two beat lines from two separate songs and then closely match them up together so they sound like a common beat. And if they get to be wrong, it can sound pretty horrible and sound like this...
(sound)
Mark - That's enough to put you off your dancing and is literally only like a few tens of milliseconds out. So, what we wanted to find out is how accurately those beat lines need to be matched for people to start moving in time to them as if they were a single common beat.
Hannah - How did you study that then? Did you go out to lots of clubs and start measuring people's dance moves?
Mark - It wasn't quite that exciting. So, we did a lab-based experiment where people basically had to just tap along to 2 metronomes played at the same time. So, what we did with those metronomes is vary the separation between them and also, the consistency and the predictability of the beats of the separate metronomes. So, we've probably got some examples I think...
(sound)
So, this is a consistent metronome where we just separate the two beats apart.
(sound)
And then this is an inconsistent metronome where the beats are no longer predictable. So, what we found was that if the individual metronomes are very consistent then they had to be very closely matched in time. So, there's not much separation between them for them to be considered as a common beat whereas on the other hand, if they're inconsistent and less predictable, then the separation could actually be larger and we still would consider them a common beat.
Hannah - So, does this mean that good DJs can mix common beats together in a seamless way, but if you're not quite so talented then you should either possibly use the fade button or try and use more irregular beats so that maybe our maybe our brains won't notice or perceive the difference?
Mark - Possibly. I mean, what we're showing is that because DJs would tend to play a very regular strong common beat with a strong beat that's very predictable then they've got to get it very accurately aligned to convince the audience that they're dancing to a single common beat when they mix it. So, they've actually got quite a hard task.
Hannah - So, there's obviously some very important applications of your research on the dance floor, but what about elsewhere in the world?
Mark - Yeah. So, we're now applying our model to quite a different situation where we're looking at synchrony within crowd movements. So for example, if you think of a football crowd, in a stadium if they get excited, they tend to all jump up and down together. And this can actually be quite a problem for structural vibration in the stadium. So, what we're looking at now is how and when people start moving together. It's kind of a similar situation because these people moving together, they're effectively defining a common beat between them. But rather than just combining sound, here, they've got a vision and maybe touch from looking at the other people around them. What we're looking at is how do they use all this conflicting information from across all different senses to actually quite simply make a common beat between them and do spontaneously start jumping up and down together.
Hannah - Thank you very much for joining us. That was Mark Elliott from Birmingham University and that study was published this week in the Proceedings B of Royal Society.

04:47 - The Longitude Prize
The Longitude Prize
with Astronomer Royal, Professor Martin Rees, Cambridge University
This week saw the relaunch of the longitude prize which was originally set by the British  government 300 years ago to encourage inventors to find a way for ships to pinpoint their positions on long ocean voyages.
government 300 years ago to encourage inventors to find a way for ships to pinpoint their positions on long ocean voyages.
It was relatively simple to work out a ship's latitude by looking at the position of the sun in the sky. But longitude, which is how far around the Earth's surface the ship was, was much harder to figure out. This prize led to a new method being developed to make determining longitude much more accurate
This new prize offers £10 million to individuals who can solve 6 pressing modern day problems.
Kate Lamble went to see the chair of the committee, astronomer royal Professor Martin Rees who started by explaining the original longitude prize...
Martin - Well in 1714, the government passed a Longitude Act and this allowed them to offer a reward of up to 20,000 pounds, which is like several million in today's money, for someone who found a way to determine longitude accurately, and specifically this had to be within 30 miles accuracy after transatlantic voyage.
Kate - And who solved the problem in the end?
Martin - Well, the interesting thing was that the solution came from John Harrison who was a real outsider. He was a Yorkshireman, started as a carpenter and joiner, and he over 40 years developed more and more precise chronometers. He won with an amazingly precise instrument, a pocket size clock which was robust enough to survive transatlantic voyages. At that time the prize was setup, it wasn't clear that clocks where the route at all. They established what many astronomers thought that it would be solved by a precise measurement to the moon and the stars. So, he was an outsider and it was a good example of how, by having a challenge prize, you draw in outsiders and allow a variety of approaches to play against each other.
Kate - I love the fact that the first reason of him being an outsider that you mentioned was being a Yorkshireman, never mind the joiner or carpenter. The Yorkshireman comes first. So, if it's a good way of bringing in outsiders why do we want to renew the Longitude Prize today?
Martin - Well of course, the scene is very different today. There's not just one big challenge but many and of course, there's a huge amount of incentives being offered by commercial bodies and other government bodies.
In fact, the 10 million pounds may seem a lot, but it's a thousandth of what the UK spends annually on our R&D. So, it's not going to change the world, but it is going to make a distinctive contribution because a well-configured prize has a number of benefits. First, it would allow lots of people to participate including some outsiders. Secondly, it can attract public interest in the progress towards the goal. One has to configure the prize so that it's challenging but not impossible. Also, the judging is objective, so it's important that the judging should be objective like in athletics, unlike the Oscars or the Turner Prize for instance.
Kate - So, if longitude was the big problem back in the day, what are the problems that we need to tackle with prizes today?
Martin - There are very many of them and I've been chair in the committee which has been picking out six areas which we think are socially important and where we think a prize with an incentive of 10 million pounds could make a difference.
These areas are:
- First antibiotic resistance, ways in which for instance someone in Africa can decide cheaply and accurately whether they've got a viral disease or bacterial disease.
- Looking after patients with dementia.
- Looking after people who have handicaps in mobility using advanced robotics and other techniques.
- Food for the world, new techniques to enable us more readily to feed 9 billion people.
- Clean water for the world. Many people are in places where they need to purify water or desalinate water.
- In the transport area, perhaps a way of moving towards zero carbon flight.
So, those are the six areas and what's going to happen is they have been announced in a TV programme last Thursday - Horizon programme and that opened the voting. Anyone can vote and the results of the vote will be announced on the One Show on the 25th of June.
And then what will happen is that our group will focus on the favoured challenge by the public and try and configure a prize. It's got to be a prize which is of the right level of difficulty, etc. So, that will be a job for us over the summer. And then the timescale for the prize would be not the 50 years that it took to solve the Longitude Prize, but something like 5 years and we would hope for there to be intermediate steps along the way so that the public can follow progress.
Kate - The original prize was solved by an outsider. Are outsiders really going to be able to help with antibiotic resistance and robotics, and more complex things we're dealing with nowadays?
Martin - Well with the example of antibiotic resistance - clearly, drug development is not the kind of thing that one is going to incentivise by this prize. But clever, simple bits of equipment which may be in conjunction with mobile phone, allow people to diagnose what sorts of disease they've got. And as regards to outsiders, anything involving software for instance can be done by very many people. It doesn't involve big bits of equipment. So, it's very important that challenge should be something that isn't going to be limited to the big battalions.
Ginny - I better start thinking about my 10 million-pound idea then. That was the Astronomer Royal Martin Rees and you can help decide which of the 6 topics gets chosen by visiting www.longitudeprize.org/your-vote.
Hannah, what do you think you'd vote for all of those?
Hannah - I probably should vote for either the paralysis or the dementia because my background is in neuroscience. However, controversially, I think my vote would be cast in more looking at sustainable food for the world and also, clean water supplies.
Ginny - I think if you want to make a difference to the biggest number of people, then water has to be the most important thing because it's just so vital. But then if I'm thinking more personally, the things that scare me about my future, it is dementia and its antibiotic resistance. So, it's a bit of a conundrum for me.
Hannah - One of the questions is whether 10 million pounds of Grant payer's money, whether this is a good idea to ask the taxpayers to help vote and to get involved in research in this way. As Martin said, this is only 0.1% of the total amount of money that goes to R&D set aside by the government. So, in some ways maybe it would be nice if we did these types of challenges or public awards more often?
Ginny - I think anything that gets people more involved in the process of science has got to be good.
Hannah - Thanks, Ginny.

10:54 - World's Largest Dinosaur
World's Largest Dinosaur
This week, a farm worker in Argentina stumbled upon what has turned out to be a bone belonging to the largest known animal to have ever walked the planet. Here's your Quick Fire Science on the find... 
- The creature is a type of dinosaur known as a sauropod, and related to the previous contender for the 'biggest dinosaur' title, the Argentinosaurus.
- Sauropods like this new dinosaur and it's famous relative the Diplodocus, had large bodies, long necks and tails, and very small heads.
- The newly discovered sauropod is thought to be 40m long and 20 m tall - the height of a 7 story building.
- It weighed 77 tonnes which makes it as heavy as 14 African elephants.
- The animal was a herbivore, and lived during the late Cretaceous period - 100 million years ago.
- Scientists think that sauropods grew so big because being bigger may have helped them fend or scare off predatory dinosaurs.
- They had long necks which allowed them to reach many different food sources to fuel their large bodies, and light bones which helped them to overcome structural constraints.
- This discovery is particularly important as they have found 150 bones, from 7 individuals, which are said to be in 'remarkable' condition.
- Previously, researchers have only had a few bones to go on, making it difficult to estimate the size and weight of the creature.
- The weight of the previous contender, Argentinosaurus was revised down from the initial estimate of 100 tonnes to 70, because estimating weight from so few bones is unreliable.
- It can also be difficult to work out the dinosaurs' proportions and overall shape when not all the bones are present.
- This dinosaur hasn't yet been named- but researchers have said the name will honour the region it was found, and the farm workers who made the discovery.

13:05 - Dengue World Cup
Dengue World Cup
with Dr Rachel Lowe, Catalan Institute of Climate Sciences in Barcelona
Ginny - Now, I don't know if you're a football fan. I must admit, I'm not, but even I  can't escape talk of the upcoming World Cup in Brazil. Conditions there are predicted to reach fever pitch for more than just the matches though because a new disease forecasting system is predicting epidemics of dengue fever. Dengue is a mosquito-borne viral illness that causes high fevers and severe bone and joint pains. In some cases, it can even be life-threatening. Dengue is endemic in Brazil and it follows a seasonal pattern linked to the weather with conditions that favour mosquito breeding, making an epidemic more likely. Now Rachel Lowe and her colleagues at the Catalan Institute of Climate Sciences in Barcelona have designed a computer model that can forecast 3 months ahead the likelihood of dengue epidemics in a given geography. Rachel gave Chris Smith the details...
can't escape talk of the upcoming World Cup in Brazil. Conditions there are predicted to reach fever pitch for more than just the matches though because a new disease forecasting system is predicting epidemics of dengue fever. Dengue is a mosquito-borne viral illness that causes high fevers and severe bone and joint pains. In some cases, it can even be life-threatening. Dengue is endemic in Brazil and it follows a seasonal pattern linked to the weather with conditions that favour mosquito breeding, making an epidemic more likely. Now Rachel Lowe and her colleagues at the Catalan Institute of Climate Sciences in Barcelona have designed a computer model that can forecast 3 months ahead the likelihood of dengue epidemics in a given geography. Rachel gave Chris Smith the details...
Rachel - We've put together an early warning system for dengue fever in brazil which brings together many different risk factors for dengue and climate information. What we then do is use forecasts of the climate information to try and predict where it may be more likely to have dengue epidemics.
Chris - This is of course relevant because pretty soon, FIFA estimate more than a million people are expected to descend on Brazil for of course the World Cup.
Rachel - Exactly. So in this case, we got together with the climate services in Brazil and the Ministry of Health, putting together the latest data on the dengue cases and the temperature and precipitation for the month preceding the games to try and say when and where there might be more likely to be dengue outbreaks or not.
Chris - So, how have you used the information we have on what's happened in the past in Brazil to try to make these predictions about where there'll be activity around the country?
Rachel - We took the dengue cases over the last 14 years for 553 regions of Brazil, combined this with information on climates and also non-climatic factors. We combine this into a model and then we used severity thresholds that are used by the Ministry of Health to work out the chance of dengue cases exceeding medium and high levels. We looked at the performance of the early warning system on past data and using this, we devised optimum trigger thresholds to indicate whether low, medium or high warning should be issued for the 12 host venues.
Chris - I see. So, by using data of what has happened in the past and how those weather patterns and so on coincide with the sorts of numbers you get for dengue, you can then have a sort of system that will enable you to predict, if the same climate patterns come along this year, this is where we would expect to have activity of the following amounts in these different geographies.
Rachel - Exactly. The model takes into account differences in dengue throughout the year and also, in different ecological zones.
Chris - When would the dengue season normally be in Brazil?
Rachel - Mainly between January and May.
Chris - So, by the time most of the people would be visiting, we would expect the disease activity to be waning.
Rachel - Yeah, we'd expect it to be exactly on the decrease.
Chris - But what does your model say about the different areas where the different parts of the World Cup are going to be played?
Rachel - In the 3 north-eastern cities of Natal, Fortaleza and Recife, there is an increased chance of a dengue outbreak. There are medium levels in some of the other stadiums such as Rio de Janeiro, Salvador and Manaus, and in the southern cities, the risk is most likely to be low.
Chris - Can you just tell us, what does a high risk mean?
Rachel - The way we defined it was a probability of dengue cases within the micro-region exceeding 300 cases per 100,000 inhabitants.
Chris - What's the likelihood of those 3 areas - Natal, Fortaleza and Recife are going to have those sorts of numbers?
Rachel - The probability of exceeding that threshold in Natal is 48%, in Fortaleza 46% and in Recife 19%.
Chris - So that's really a reasonable chance of high dengue activity in 3 areas which do overlap with where games are scheduled to be played.
Rachel - Yes, so the idea of this model is, it's capable of issuing warnings 3 months in advance. That can be incorporated into the decision making of the local authorities and the dengue control people to help concentrate intervention activities in those areas.
Chris - Would that mean for instance spraying for mosquitoes, giving people advice, telling people to wear mosquito repellents for example.
Rachel - Yes and the dengue control teams have been going house to house, checking for potential mosquito breeding sites, destroying them.
Chris - What do the Brazilian authorities say about this? I presume you've shared your findings with them.
Rachel - Yes. We actually are, our Co-authors included scientists from the Ministry of Health. They've been very supportive of the study. They provided inputs about how to use these results in the health policy way and they provided us with the latest data in order to produce the forecast.
Chris - But more importantly, have they also now formulated a strategy for the World Cup based upon your predictions which suggests that there are 3 hotspots which fans maybe need to be aware of and which authorities need to act on.
Rachel - The National Dengue Control Programme are working very hard in all the cities ahead of the World Cup to make sure that the dengue risk will be at the minimum.
Ginny - Dr. Rachel Lowe from the Catalan Institute of Climate Sciences in Barcelona.
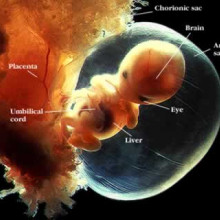
18:24 - Placentas teeming with bugs
Placentas teeming with bugs
with Kjersti Aagaard, Baylor College of Medicine in Texas
Hannah - Now, sticking with the subject of bugs, we don't want to put you off the 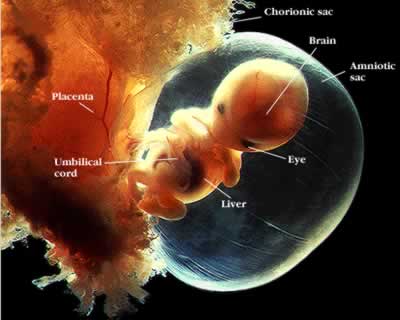 football on your tea, but our bodies are teaming with trillions of bacteria anytime. In fact, 9 out of 10 cells in your body are bugs. As I called 'microbiome', it's usually made up of friendly bacteria that live in the gut on the skin in the mouth and elsewhere, helping to keep us healthy. Billions of bugs are already on us and inside a baby within the first few days of life. But where do these bugs come from? Kjersti Aagaard from the Baylor College of Medicine in Texas thinks that she's found the answer and it's not what you might expect. Kat Arney spoke to her to find out more.
football on your tea, but our bodies are teaming with trillions of bacteria anytime. In fact, 9 out of 10 cells in your body are bugs. As I called 'microbiome', it's usually made up of friendly bacteria that live in the gut on the skin in the mouth and elsewhere, helping to keep us healthy. Billions of bugs are already on us and inside a baby within the first few days of life. But where do these bugs come from? Kjersti Aagaard from the Baylor College of Medicine in Texas thinks that she's found the answer and it's not what you might expect. Kat Arney spoke to her to find out more.
Kjersti - The question we're trying to answer is, does it start at the time of birth, does it start depending upon how that baby is born, whether it's through the vaginal canal or via caesarean?
And we had a couple of clues early on that it may actually precede the immediate time around birth. So, other investigators, if they looked at the phyla, the different bacteria in an infant's gut in the first week of life, at a very high level view, they found out that half of the bacteria that are present were what we call actinobacteria which includes bifidobacterium and propionibacterium. And then there were smaller minority that were bacteroidetes and firmicutes. Included in this firmicutes is lactobacillus. What we were intrigued by is that very small 10% to 20% or so that are present in that infant in that first week of life, lactobacillus, were really the only ones that are dominant in the vaginal flora. In other words, some 80% or more of the different types of bacteria that populate that infant at the time of birth or within that first week of life weren't coming from the vagina.
Kat - Now, that's very controversial because we sort of have this idea that as baby comes out of mum, it gets covered with all these bacteria and that's what colonises it. Where have these bugs come from?
Kjersti - We said there were kind of two things that weren't making a lot of sense to us. So, one was what I'd call the science math which was, what was present in that infant's gut didn't seem to match real well what was present in the vagina. In addition as a clinical obstetrician, babies don't spend a lot of time hanging out in the vagina on their way out of the uterus and into the world. They're moving through it pretty quickly. So, how is it exactly that they would acquire all of these microbes, simply as the passage through the birth canal? So, we were curious whether it was possible that first of all, the infant wasn't that sterile at birth and whether or not it was actually being exposed even in normal non-infective settings prior to delivery.
Kat - Where are these mystery microbes coming from?
Kjersti - Now, when we were just starting to complete this study, another group here in the US showed using very traditional microscopic techniques that if you looked in the placenta, nearly a half, just over a third or so, had evidence of bacteria in completely normal, uncomplicated, non-infected placentas. So, we kind of took that up and said, "Great! We're going to use a metagenomic or DNA based microscope. We're going to look as detailed and as robustly as we possibly can within that placenta and see what actually is there and what they're capable of doing."
Kat - And so, what did you find?
Kjersti - So, what we found was that first of all, there were bacteria present in the placenta. Now, they're what we call low biomass which means that the placenta isn't teaming with that bacteria. If we had to make a rough calculation, we'd say that for every pound or approximately 500 to 600 grams of placental tissue, there is about a gram of bacterial DNA. We don't really know what that equates to per number of cells because we haven't figured that out yet, but that work is underway and we may get a better appreciation for it. But when we looked at who was there, what we found when we compared it to other microbiome body sites is that placental microbiome started to look the most like the oral microbiome and didn't actually look very much like either the gut or the vagina. It really looked like the mouth.
Kat - Now, this is just getting weirder. So, how does the kind of bacteria that are in the mouth get into the placenta and then how do they get from the placenta into the baby?
Kjersti - We don't know exactly how they get there. We can make some educated guesses as to how we think they get there and we think they're primarily spread through the blood. What we think is that some of the bacteria that are in the mouth actually do a very good job of making their way through the endothelial linings in blood vessels, and potentially then pave the way for other bacteria like E. coli to make their way.
Hannah - That was Kjersti Aagaard from the Baylor College of Medicine in Texas. So Ginny, bearing that in mind that there's this amazing biome in the placenta, if you gave birth, would you eat your raw placenta or maybe offer it to your boyfriend as a present? I don't know.
Ginny - I must say that's something I've never actually considered before. It's a bit gross. I'm first kind of thinking but I guess if there was some strong scientific evidence that it would provide a benefit then I might consider it. But I think I'm going to wait until there'll be a few more studies before I say yes for sure.

24:02 - Pricing the value of life
Pricing the value of life
with Carole Longson, National Institute for Health and Care Excellence
 Hannah - First up, in the UK our National Health service, as we hear in the newspaper all the time, has limits on its budgets. And that means we have to decide which drugs we can afford to fund and which are out of our price range.
Hannah - First up, in the UK our National Health service, as we hear in the newspaper all the time, has limits on its budgets. And that means we have to decide which drugs we can afford to fund and which are out of our price range.
So how do we decide which drugs we have access to on the NHS?
NICE or the National Institute for Health and Care Excellence, are the body that makes these recommendations after balancing the number of years of life that would be added due to a medicine and the quality of that added life with the cost of the medicine to come up with a figure known as a QALY.
Kate Lamble asked the Director of their Centre for Health Technology Evaluation, Carole Longson how they come to a decision.
Carole - We could talk about this in a very technical way. We use health economics and cost-effectiveness analysis. So, they're the technical terms but in essence, at the heart of it is, trying to understand the added benefit that a medicine or other technology might bring and compare that to the added costs that we have to incur when we're using that. that's called cost-effectiveness analysis. But it's very much like looking at a budget and thinking about the best way to spend the money that they have available to deliver the best effects.
Kate - So, when you have to know how effective the drug is, who gives you that information?
Carole - Increasingly, we're taking a look at medicines very early and as a consequence of that, we rely quiet heavily on the companies that produce those medicines to provide the evidence to us. And added to that, we ask patient groups, we ask health professionals to give us information and an understanding and experience of what it's like to both live with that condition and also, manage that condition in the healthcare service. It's all that information that comes into our decision-making which is done by an independent committee. It's not NICE staff that sit on that committee. An independent committee that takes a look at all of that evidence and information in the round when judging whether or not a new medicine is in fact, good value for money.
Kate - Some people like for example the science journalist Ben Goldacre argue that there's a bias in the evidence that some pharmaceutical companies produce towards positive results and negative results that show it's not that effective, aren't necessarily brought up. Do you think that that can in any way influence NICE's decision?
Carole - So, what Ben Goldacre is talking about there is what the researcher's term, publication bias, it's actually a very standard common phenomenon in science overall. So, it's not just about the companies. In that researchers tend to want to tell people about their positive results. When the results are not so positive, tend not to want to publish. And you do have to be very careful that you're looking at all of the data. We would use both published and non-published data, but you're right. it is a very, very important aspect of all science to be honest that you are careful about the data that you are looking at and you make inferences about what data may or may not be available at the time that you're looking and making the decision.
Kate - So, you take into account evidence published and unpublished, but also these personal accounts of what it's like living with a disease for example. How do you make that idea of this cost-benefit analysis? Is any amount too much?
Carole - This gets into the area of, again, an economic term called 'opportunity cost'. You've got a pot of money and if you use it to buy something, you can't use it to buy something else. You can only spend the money once. And through some analysis, we can understand what it is, the health service in particular is using their budget for and also, how much health they're generating for that spend. We use that as the basis of estimating value for money. That opportunity cost translates into something called a cost per quality adjusted life year so another technical term. And at a particular level of a quality adjusted life year which is 20,000 pounds per quality adjusted life year, we know that the NHS gets good value for money at that level.
Kate - And so, this 20,000-pound figure, when we're talking about a quality adjusted life year, are we saying that if I were sick, that would be a good year of my life.
Carole - What we're saying is that a new technology can bring improved quality of life. So, how well you're able to undertake activities of daily living for example or extend your life, or hopefully, both of those things. So again, it's not about the cost to gain that particular increment. It's about how that increment stacks up against how much you have to pay to gain it. That's really at the heart of what we do. It's not the whole thing that we take into account because all of those patient accounts and thinking about the efficiency of the healthcare system also feeds into the judgement which in the end, the committee has to make it. So, it's not an algorithm. It's not a flowchart for how to do this. It's a judgement.
Kate - So, NICE sort of acts as the gateway to the NHS which is quite a big market for drugs I suppose. How much is their - you're the doorman sort of guiding the way and they have to be effective and how much is their some kind of negotiation about the price or can they come back to you if something is rejected?
Carole - Now, that's a really interesting question. Yes, we probably do see our role, both as - I suppose as gatekeepers and enablers. So, we do have to be very careful that the NHS in using new medicines is spending that money as wisely as they can. But we have to do that in a way that enables these promising new innovations to be used in our healthcare system. Recently, it's been introduced a concept of patient access schemes. This provide a mechanism for companies to reflect and come back to us with a new proposal and they have provided an extremely useful mechanism for us to act in that enabling role with companies.
Kate - When you reject a drug, there was a recent 90,000-pound breast cancer drug that was rejected and it sort of hit the headlines because there are a lot of patient groups who obviously feel like they need the drug and they get upset that it wouldn't be made available to them. Did you at NICE understand their position and why they're so desperate? Does it become very difficult for the people on the panels to make those decisions?
Carole - The people on those panels really do have probably one of the hardest jobs in public life. But what they really do have to do is to try to balance those needs of the individual with the needs of everybody who uses the NHS and to make sure that their judgments reflect to that balance as best they can.

31:24 - Why are drugs so expensive?
Why are drugs so expensive?
with Jennifer Cohn, Medicine Sans Frontiers, Joshua Cohen, Tufts University
 How do developing countries gain access to treatments? And why do drugs cost so much in the first place?
How do developing countries gain access to treatments? And why do drugs cost so much in the first place?
Ginny - We use NICE to help us decide which drugs we should have access to, but what do other countries do, and how do we make sure that low and middle income countries still have access to live saving medicines?
Jennifer Cohn is from Medicine Sans Frontiers and Joshua Cohen is from Tufts University in the US.
Joshua - I think it depends very much on the political lens through which Americans may look at something like the National Institute for Clinical Excellence. Those in the US opposed to government regulations and rationing would think of NICE as a death panel of sorts and those who are in favour of some degree of regulations by government would view NICE as a useful framework. However, it's not directly translatable to the US healthcare system. The system we have in the United States is decentralised to a degree that certainly, the UK is not. With literally hundreds of payers and 50 states and federal state division of labour with respect to government, you're not going to see a NICE-like body being instituted. That being said, Obama Care in some ways does use NICE-like mechanisms such as spending lots of money on comparative effectiveness research.
Ginny - So, that's a good point. In America or in the UK, we do actually have a reasonable amount of money to spend. So, when we're looking at how expensive drugs are, we have a very different framework from say, low and middle income countries. Jennifer, I know you work with those sort of countries. Do they pay the same sort of amount as us to drugs?
Jennifer - It really depends. So, many low income countries can have access to generic medicines and some cases, before countries like the US or UK can. And that's because of laws that recognise that public health needs may come before patent and profits. Now, that's not to say necessarily that these drugs are always affordable. However, let's take the example of HIV. In 1999, the cost of a brand name first line anti-retroviral regimen to treat HIV in low and middle income countries was about $10,000 per patient per year. Now, that's far, far above what most countries could afford and this again is at a time when countries in Southern Africa such as Swaziland and even Botswana were looking at HIV infection rates of about 40% or nearly 1 in 2 people. Now, within about a year, prices came down dramatically from $10,000 per patient per year to about $350 per patient per year. And why it was that? that was almost exclusively because of the entry of generic manufacturers that produced quality versions of these exact same medicines for patients. And the effects of competition can rapidly bring down prices and thus increased access to patients who desperately need these drugs.
Ginny - Jennifer, when we're talking about low and middle income countries, is it just places like Africa or are there other places in EU that are paying less than us for drugs?
Jennifer - Absolutely. So, in many cases, the drugs that are available generically may not just be available in Africa. So again, for instance, countries like Thailand may have access to generics as well. Countries in the EU generally follow the same patent laws. In fact, the EU does have an agreement that lowest cost medicine in one country in the EU can actually be imported to other countries in the European Union. But that's just an agreement within the European Union.
Ginny - Now, I've had a question in from Facebook. Richard Lyle says, "What can be done to reduce the cost of bringing a new drug to market without putting patients' safety at risk?" So drug companies tell us that they have to charge these high prices because of all the development cost and we actually asked several drug companies to come along and talk to us tonight. Unfortunately, none of them were available, but Joshua, what do you think? Could we bring the prices down?
Joshua - Yes, but let me first start out by saying, the high cost of drug development is not and should not be a justification per sey of high prices. Markets may justify high prices if the drug is highly valued by patients and doctors. Especially if we're talking about brand name drugs - drugs for which they are limited or no treatment alternatives. In a competitive market, price should be a reflection of supply and demand. And in particular, an informed consumer's willingness to pay for a drug. But the problem is, healthcare markets are notoriously characterised by some degree of market failure. And the most distinctive feature of this market failure is the fact that consumers are generally not well informed about a drug's value or its cost. This is really where NICE and other health technology assessment organisations also in the US would come in and make some kind of value assessment of the drugs that are in the market. Now, we should distinguish between the brand name drugs, new drugs, and the particular drugs that create a unique therapeutic class from generic drugs. In the United States, about 85% of prescriptions are now generic. The pricing debate certainly does not affect generics. It's not about generics. It's about those brand name drugs targeting populations, often orphan populations. The drugs are quite expensive and that's really what the pricing debate is looking at.
Ginny - We'll have to leave it there I'm afraid, but thank you so much to Jennifer Cone and Joshua Cohen for joining me.
37:19 - Hacking your DNA data
Hacking your DNA data
with Yaniv Erlich, MIT
Hannah - This week we've been talking about ethical issues in medicine and so far we've  been discussing the price of drugs.
been discussing the price of drugs.
But in recent years medicine is becoming more and more personalised, with drugs being adapted to our specific DNA codes.
With all this sensitive data needing to be stored some people including Yaniv Erlich from MIT concerns about the safety and privacy of our genetic information as he explained to Kate Lamble
Yaniv - The genome is basically 3 billion letters that consist our DNA. This is the material that is inherited from our parents and basically represents our heritage. For most of the traits and the diseases that we know, there's a strong genetic component. But it's important to emphasise that for most of the diseases, the genome doesn't determine the trait it just increases the predispositions. By analysing genomes, we can understand this tendencies in the hope that in the future, we can suggest treatments based on the genetic makeup of the person, what is called personalised medicine.
Kate - So, you're concerned about the security of this genetic data. If I gave my genetic data to somebody, what would be the problem if someone identified me? I mean, I put lots of information on Facebook and Twitter, in the public domain about myself all the time. how would someone having my genetic information be different? What could they do with it?
Yaniv - The thing is that, it's a question of personal taste. Some people are really open about showing data about themselves. Other people have some concerns about their privacy. Some people are concerned that they might be discriminated based on their genetic makeup. So, our project was mainly to show that this can happen and to tell people that they should be aware of that.
Kate - Are there laws against it? As well as just protecting ourselves sort of by not sharing our data, are there laws being enacted to prevent this from happening in the future?
Yaniv - So, here in the US, we have GINA - the Genetic Information Non-discrimination Act. According to GINA, you cannot be discriminated in your workplace or by health insurance based on your genetic material. But GINA as of today doesn't protect you from discrimination by your life insurance company or long term care. So, it gives you some cover, but the blanket is not full. There are few services that you'll be denied based on your genetic makeup.
Kate - If I donated my genetic data or gave my genetic data to either research institute or to my doctor for some personalised medicine, they were going to anonymise that data, take my name off it. How could somebody then know that that data was mine?
Yaniv - Your genome is a strong identifier of yourself and I will tell you a little bit about our study. We focus on males. Now males get the Y-chromosome from their father and therefore, they got his Y-chromosome from his father. Now, in most western societies, males get their surnames from their fathers as well. So, this creates a correlation between the Y-chromosome and the surname. What we showed that you can take whole genome sequencing data that is apparently de-identified or anonymised and then by inspecting the Y-chromosome and searching it online genetic genealogy databases, you can find the surname of the individual. And then we considered if you know the age of the person and the geographical location. In most cases, you have this information as part of the metadata. So, we said, "Okay, if you have surname, you have the age, and you have the geographical region, you can really identify the person and get to this individual." We showed that in several examples that we can get to people that posted their genome anonymously on the web that we can identify them.
Kate - It amazes me that these genetic databases that you can use to identify somebody's surname and just lying around on the web. How did we get ourselves into a situation where that information is just out there?
Yaniv - Many people are interested about their heritage and about their ancestry. This databases created by the really vibrant community of genetic genealogists - by the way, I'm part of this community as well. They also put my Y-chromosome and my surname in these databases because I wanted to know more about my heritage. Now, the thing is that, if you contribute, it enabled people to identify all your patrilineal relatives.
Kate - So, if we want to protect this data and do more than just anonymise it, I mean, I share my banking details online to buy things on the internet through PayPal and things all the time. can we just use the same practices that they do to protect our genetic information?
Yaniv - It's a bit different because when you share your bank information, what you're trying to protect is from a third party that will tap into the conversation or the online transaction. But the bank that you are communicating with sees all the information. So, we cannot use just the simple encryption methods, but there are new methods that are currently developed but I have to say that they're still in their infancy. In these methods, you could share genetic information. Let's say, we have some companies that try to interpret the information for you. And under these encryption methods called homomorphic encryption, the interpretation company, the company that tries to say what are the predispositions that you have, they don't really see the actual data. So, think about that you have a brick of gold and you want to go to a jewellery maker, but you don't really trust that jewellery maker. He might just take the brick of gold and run away. So, what you could do, you could take this brick of gold, place it in a glove box and put inside the glove box all the equipment that he needs to make the necklace. You lock that glove box with the best lock in the world and now, you give it to the jewellery maker. Now, the person has not interest of finding a way because he will never be able to break into the glove box and steal the gold brick, but he can still make the necklace for you. The same way this encryption method that you put your genetic information inside this mathematical glove box. Now, you give this glove box to the company. They know how to process this encrypted data and to derive the predispositions, but they don't see the actual results. They cannot just take them and run away. You get back this glove box, you open with your key, and now, you get these processed genetic information that you can do something with it. You can go to your physician or use it for other purposes.

44:29 - Selecting babies by their DNA
Selecting babies by their DNA
with Julian Swavelesco, Director of the Oxford Practical Centre for Ethics, Amy Taylor, genetic counsellor at Addenbrookes Hospital
 How genetics is being used to screen embryo's for disease, could couples create designer babies, and should the government interfere?
How genetics is being used to screen embryo's for disease, could couples create designer babies, and should the government interfere?
Julian - Yes, using IVF and genetic diagnosis, you can test embryos at the moment legally for dispositions to major diseases. So, chromosomal diseases like Down Syndrome or single gene disorders like cystic fibrosis, thalassemia, Huntington's disease and some early onset forms of Alzheimer's disease. But you can only test for the genes which are associated with what the Human Fertilisation Embryology Authority judges to be serious diseases, you can't for example test for the sex of the baby and choose to have a male or female embryo and nor can you test for genes that are associated with various abilities or personality traits.
Hannah - Thank you, Julian and I suppose previously, the situation was that if a mother was at risk of having a child with Down Syndrome for example, she might have an amniotic fluid sample and cells and then have to face a very difficult decision as to whether to continue with that pregnancy or whether to abort. It is obviously a very tricky issue to face. Amy, I believe that you actually work with some of the parents who are facing these types of decisions. How do you talk about these issues with them?
Amy - Well, we see families who have a known genetic condition that's been inherited through their family. We start by exploring with them how they feel about the possibility of having a child affected by that condition. Some couples will be accepting of that. that depends on the severity of the condition, their own experience of it in the family. Some will feel very strongly that they don't wish that condition to be passed on to the next generation. So, if they take that position then pre-implantation genetic diagnosis or PGD is an option for them. It's a simpler option or it's an ethically more comfortable option for them than to go through pregnancy and have diagnosis and then have termination. But that's not to say that it's easy at all. Like all IVF, it's a lengthy process. A woman has to have her ovary stimulated to produce several eggs. It takes probably about a year from when we refer a couple to the PGD Centre which is in London to when they might have actually an embryo implanted. So, it's not something that couples undertake lightly. They do it if they feel strongly that - usually, their reasoning is that it would be unfair on a child to have that condition.
Hannah - I believe that there's a big community of people that are affected by deafness. This is another condition which is listed within the guidelines of something that we can legally, within the UK use this pre-implantation diagnosis genetic screening treatment for. There's lots of people that say that actually, deafness isn't a disability. It provides another form of language for people and we shouldn't be able to select those genes out of the gene pool.
Amy - That's true. So, as Julian mentioned, the Human Fertilisation and Embryology Authority, the HFEA decide what conditions can be selected for using PGD. One gene that causes deafness connects in 26 is on that list. The way that they make those decisions about what conditions are added to the list is dependent on how severe a condition is. They do use public consultation. So, deafness is probably at the milder end of the conditions where PGD is allowable. You're absolutely right. The deaf community feel very strongly that deafness is just a normal variation and that it shouldn't be something that you should select out of the population.
Hannah - Julian, I believe that also, as we're getting more information on the genetic basis of disorders like Schizophrenia for example or autism or attention deficit hyperactivity disorder, there's a possibility that people may want to select embryos out that have a genetic predisposition to disorders like that. Is there a worry that that might happen in the future? Are we playing God a little bit too much? Are we trying too much to shape society?
Julian - Well, you can sequence the whole genome of an embryo at the moment, but it's very expensive. It has come down to about a thousand dollars. So eventually, you'll be able to look at all the genes that are associated with say, psychiatric diseases such as Schizophrenia or depression. When you have 10 or 20 embryos and you've found the ones which don't have major genetic diseases, you'll then have a risk profile. So, embryo 1 will have a higher chance of developing Schizophrenia or embryo 2 will maybe have a higher chance of depression. It's my view that people should be free to make those decisions. The state doesn't have a role in constraining their liberty. They should have access to that information when it's available. And they should make decisions about what kind of child they think will have the best chance of the best life. I think the worry is that at the moment, you wouldn't be allowed to test for those risk factors even though the information is available.
Hannah - And, what about the fact that some of these genes might actually confer an evolutionary advantage? You've got 10 seconds to try and answer that.
Julian - Well then you have to factor that into the decision then. I think if you give people freedom, some people will choose to select against that, some people won't. You're only talking about selecting it from embryos that nature might have picked out. You're not talking about genetically engineering people.

50:36 - Is some of the universe anti-matter?
Is some of the universe anti-matter?
We take this question to Tamela Maciel, postgraduate astrophysicist at Cambridge University....
Tamela - If astronomers caught sight of a distant and isolated galaxy composed entirely of antimatter, they'd have trouble spotting the difference between it and a normal matter galaxy. That's because apart from having an opposite electric charge and magnetic moment, an antimatter particle has the same mass and internal structure as its matter counterpart. So light emitted from an excited antihydrogen would look exactly the same as from a normal hydrogen atom. And a galaxy of antimatter would have the same mass, and therefore the same gravitational force, as a matter galaxy. Antimatter is exotic neither because it is very different from normal matter nor because it follows different physical rules, but simply because it is very rare in our present-day universe.
Very high energies are required in order to produce antimatter. On earth, CERN's Large Hadron Collider routinely creates antimatter as the natural byproduct of high-energy particle collisions. The challenge is storing this antimatter for any length of time - every time antimatter collides with normal matter both are annihilated in a flash of light. And this happens a lot in our matter-dominated world. Nevertheless a few years ago physicists at CERN were able to study a newly-minted antihydrogen atom in isolation for a full 17 minutes before it eventually disappeared. In space, where much more energetic collisions occur, antimatter is also regularly detected. But usually only in small, discrete quantities arising from well-understood particle interactions.
So how do we know there aren't any antimatter galaxies lurking out there in the universe? The flash of gamma ray light that results from matter-antimatter annihilation is an excellent antimatter detector. If there are galaxies made up entirely of antimatter, then on the edges of these galaxies where the antimatter meets normal matter we would expect to see lots of gamma rays from annihilation. But despite careful observation from the likes of ESA's INTEGRAL or NASA's Fermi Gamma-ray Space Telescope, this tell-tale antimatter signature has not been observed, so astronomers believe that antimatter galaxies and clusters of galaxies do not exist.
But perhaps all the antimatter galaxies are sitting away by themselves in some distant corner of the universe where normal matter is rare. This possible separation of matter and antimatter into distinct corners of the universe must have happened immediately after the birth of the universe and before the period of rapid expansion known as inflation. But even in the high-energy soup of the very early universe, none of our current particle physics theories can provide a mechanism allowing antimatter and matter to separate spatially. Remember that in many ways, antimatter behaves just the same as normal matter and follows the same physical laws. So at the moment, it seems impossible that antimatter could have clumped and separated away from matter at any point during the history of the universe.
The biggest question in this discussion is why is there so much matter? Equal amounts of matter and antimatter should have been produced from the aftermath of the Big Bang. But with equal amounts, all the matter and antimatter should have annihilated long ago leaving only light. Instead there must have been a slight imbalance in the amount of matter produced compared to antimatter which meant that over time the annihilation process gradually left only the excess matter which forms our universe today. Despite a few potential theories, we still don't know exactly why the imbalance exists and this enigma continues to be a big topic of research.










Comments
Add a comment