From detecting tumour DNA in our bloodstreams to making cancer cells stand out in an MRI scan, this week, coinciding with the NCRI Cancer Conference, we explore how best ways to detect cancer and monitor tumours during treatment. Plus, in the news, what a dog's wagging tail can tell you about its mood, the chemistry behind fireworks, how wind farms could be made up to 30% more efficient just by moving the turbines around, and the electronic blood that could help to shrink supercomputers...
In this episode

01:37 - Tail wags the dog
Tail wags the dog
The direction in which a dog wags its tail tells other canines what it's thinking, new research has revealed.
Scientists have confirmed previously that tail  wagging isn't just a back and forth affair: the wag can be biased towards either the right or left.
wagging isn't just a back and forth affair: the wag can be biased towards either the right or left.
Dogs watching the approach of - and greeting - their owners, or other dogs they are friendly with, tend to wag more towards the right. On the other hand, a cautious dog, considering doing a runner because it's seen a potential threat in the form of a larger, unfamiliar animal, will tend to wag more to the left.
Whether dogs themselves are sensitive to the wagging signals of their canine counterparts wasn't known, however, until now.
Writing in Current Biology, University of Bari Aldo Moro researcher Marcello Siniscalchi and his colleagues showed 43 domestic dogs, of both sexes and from a range of breeds, video sequences of unfamiliar dogs describing either left or right-biased tail wags.
The team monitored the dogs' behaviours and heart rates during the process to gauge their degree of stress.
Watching tail wags to the left significantly elevated the animals' stress levels with their heart rates increasing to almost 200 beats per minute.
A non-moving / wagging picture elicited a smaller increase - to a heartrate of about 150, and rightwards wags elicited an increase to about 140.
The team also repeated the experiment with dog-shaped silhouttes to avoid any non-wagging related bias and obtained the same result.
This shows, they say, that domestic dogs actively extract communication cues from the tail wags of their peers.
The reason it happens, they suggest, is because the brain's left hemisphere - which controls movements on the right side of the body - contains a region specialised for "approach" or friendly behaviour, while the right hemisphere contains a region specialised for withdrawal or retreat behaviours.
"The finding that dogs are sensitive to the asymmetric tail expressions of other dogs supports the hypothesis of a major role of social behavior in the evolution of brain asymmetries," the team speculate.
"It also opens a window to the objective investigation of the emotional lives of animals."
04:36 - Earth-sized exoplanet, but 3000 degrees
Earth-sized exoplanet, but 3000 degrees
A planet roughly the same size and with the same composition as the Earth has been 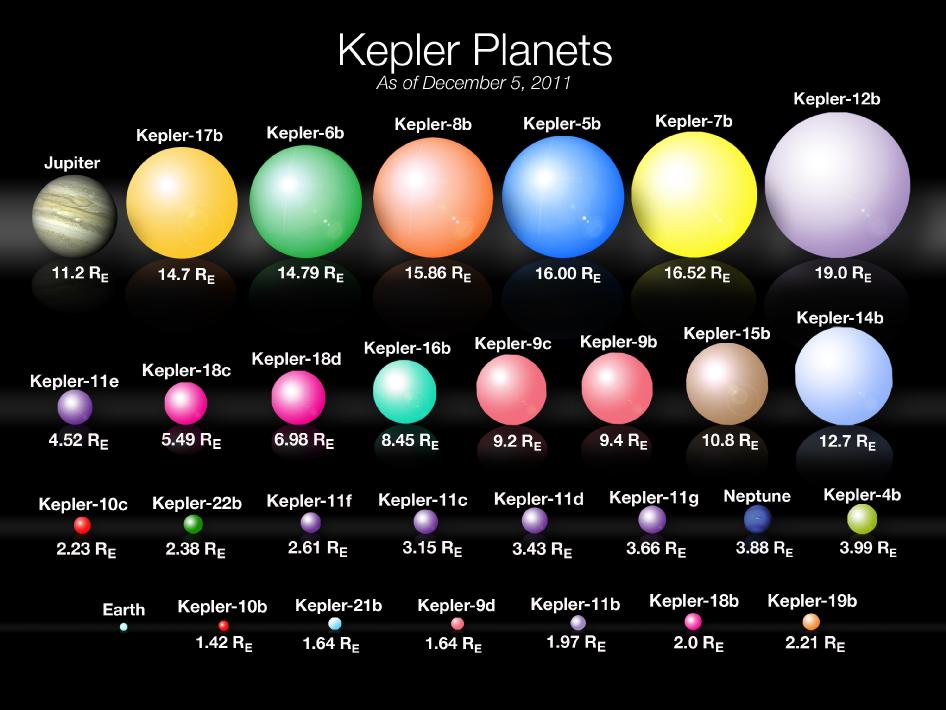 discovered orbiting a distant star.
discovered orbiting a distant star.
Since 1995, when the first so-called "exoplanet" was discovered orbiting a star other than our own Sun, a further 999 similar bodies have now been documented.
But, owing to the difficulty intrinsic to finding them - which involves laboriously looking for minute wobbles or dips in a star's light as the planet orbits - most of the logged exoplanets are enormous Jupiter-sized objects.
Now, two papers in Nature have produced one of the most exciting results yet: the confirmation of a planet that's about the same size and about the same density as Earth.
Unfortunately, that's where the similarities end, because this planet, Kepler 78b, is orbiting 100 times closer to its host star than we do, so its surface is at a sizzling 3000 degrees Celsius.
At these temperatures, there's no prospect for life or much of an atmosphere. But what's key is that the approaches taken by Andrew Howard and his colleagues at the University of Hawaii at Manoa, and Francisco Pepe and his team from the University of Geneva, prove that Earth-scale planets can be found. And, by careful monitoring of the way that the light coming from the host star is eclipsed, and also stretched and shrunk by the orbiting planet's gravity, accurate estimations of the planet's size and mass can be made.
Every 8.5 hours, corresponding to one orbital period, the team found that the intensity of the light dropped by 0.02% as the planet passed in front of the star.
The measurements presented by the two teams agree that Kepler 78b weighs the equivalent of about 1.86 Earths and is about the same diameter as our own planet.
The density is about 5.5g per centimetre-cubed, nearly the same as Earth, and consistent with a composition of 20% iron, 80% rock.
This finding makes it "the smallest exoplanet yet for which the mass and radius are known."
Although the surface conditions are hotter than a furnace, the existence of Kepler-78b shows that, at the very least, extrasolar planets of Earth-like composition are not rare.
To find more of them, NASA is preparing TESS - the Transiting Exoplanet Survey Satellite - which will spot promising candidates for closer inspection by Hubble's successor, the James Webb telescope, earmarked for launch in 2018...

07:41 - What do you need to make a firework?
What do you need to make a firework?
Remember, remember the fifth of  NovemberGunpowder treason and plot.We see no reason, why gunpowder treasonShould ever be forgot!
NovemberGunpowder treason and plot.We see no reason, why gunpowder treasonShould ever be forgot!
So here's your quick fire science on gunpowder and fireworks just in time for Bonfire Night, with Kate Lamble and Simon Bishop.
- It's often said that the Chinese invented gunpowder about 2,000 years ago. The first firecracker was a gunpowder-filled bamboo stick.
- The recipe for gunpowder was perfected in Europe in 1560. It has remained mostly unchanged ever since. To make a firework you need five ingredients: an oxidizer, a reducing agent, regulators, binders and colourants.
- Oxidizers are molecules that split apart to release oxygen. They are typically metals combined with nitrogen or chlorine.
- The released oxygen is then used to burn a fuel, called a reducing agent. This burning produces hot gases, usually sulphur dioxide and carbon dioxide.
- The oxidant in gunpowder is potassium nitrate, which releases oxygen to help burn sulphur and charcoal. Gases then build up inside a container, and when they are suddenly released - boom!
- In fireworks, these gases are pushed down at the ground, firing the rocket up into the air instead of causing an explosion. There is then a second compartment full of gunpowder, oxidants and chemicals that give out coloured light.
- The third crucial ingredient is a regulating chemical, which speeds up or slows down the reaction depending on the desired effect -- sparklers need to burn quickly, for example, and the gases push out small lumps of burning iron or titanium, creating the sparks. Fireworks use everything from metal powders to cornmeal as regulators.
- Binder chemicals hold everything together. In sparklers, common binders are sugar or food glazes, dampened with water or even alcohol.
- To create light, fireworks are packed with metal salts. Different metals burn with different colours -- burning aluminium will give white light, oranges come from calcium, and green and blue colours come from burning copper.
- It's all of these ingredients that come together to make firework displays so exciting. But the world record for the most fireworks launched within 30 seconds might take some beating. In 2010 in the Philippines, it took just 17 seconds to launch nearly 126,000 rockets....
We hope you enjoy Bonfire Night fireworks this year! Just remember to stay safe - always read instructions, remember to stay well back, and never go back to a lit firework.

10:23 - Designing better wind farms
Designing better wind farms
with Cristina Archer, University of Delaware
Driving through the countryside you often see huge grids of wind turbines  dotted across the fields. But does it actually matter how we lay them out? Cristina Archer, from the University of Delaware, has been studying how air flows through wind farms in order to answer this question. She spoke to Chris Smith.
dotted across the fields. But does it actually matter how we lay them out? Cristina Archer, from the University of Delaware, has been studying how air flows through wind farms in order to answer this question. She spoke to Chris Smith.
Chris - So first of all, what sorts of improvements could we make then by changing the way that we format where the windmills are in these aggregations of wind farm?
Cristina - Well, my research found some staggering results actually if I can make this joke. You can actually improve the performance of a wind farm by 13 to 33% if you place your turbines in a staggered arrangement. What I mean by that is, it's easier to describe if you think about yourself at a movie theatre and you want to have a good vision of the screen. And of course, you don't want to have a person sitting right in front of you. And so, the way turbines are often arranged are kind of like the seats in a theatre. They're like aligned with one another which is not very smart because if you want to look at the screen, you will like the person in front of you to not be just exactly in front of you, but slightly staggered.
Chris - Why do you think that people chose to put them in the theatre arrangement when they didn't really have to do that? Was that just our obsession with order?
Cristina - I know. Maybe it is. If you have an event in your yard, say, a wedding or something, you will see that the natural instinct of us humans is to make things lined up. It's just easier you know, so it's very understandable.
Chris - The numbers you're suggesting of between 13 and 30% improvement in the productivity of a wind farm. Those are enormous numbers! How did you do this and why did you begin looking at this?
Cristina - First of all, the ultimate goal is to try to reduce the cost of wind energy. Wind energy is really relatively cheap compared to all the other renewable energy sources. It's possibly the cheapest. But the designs of turbines are already very good - the blades are optimised, the airfoils are great, they're even beautiful at this point. So, there's not really that much you can do with the design of turbines to make them even more productive. They're already pretty much as efficient as it gets. And so, if you want to reduce the cost of wind energy, you have to act on something else. What I was interested in is, can we place them in such a way that the wakes that the turbines generate - you can think of a wake as turbulent eddies - once that wake hits the next turbine, obviously, it impacts its productivity. So, you don't want these wakes to interfere too much with the turbines that are downstream.
Chris - So, you're saying, instead of the view in the cinema being occluded by the head of the person in front, you don't want dirty air which is all messed up and full of turbulence, hitting the turbine behind. You want to minimise that effect. So, how did you actually do that? How did you try and work out what the effect was and how best to place them?
Cristina - I used a very sophisticated simulator. This is a computer code that tries to resolve the equations of motion of the atmosphere. Very fancy terms to say that I'm trying to basically predict what the wind will be in all the points, millions of them, in a wind farm. So, you have to imagine like a cube that includes the entire farm, and I'm trying to predict exactly what the wind speed, what the temperature, what the pressure will be at all these million points. I have include obviously the presence of the turbines in this volume, the fact that they rotate and the fact that the turbulence generated by a turbine can impact the following one.
Chris - You must have had a very powerful computer to do that.
Cristina - Yes, absolutely. Actually, it's not even powerful enough. I would like more power. If any of the listeners have some kind of a super duper, even more powerful system, please let me know. Right now, I'm using a high performance computer cluster with over 200 parallel processors and it's still going to take me about 2 months per simulation.
Chris - Going to have to open a few wind farms to power that, aren't you? So before we finish, could you just tell us then, have you actually tested this and validated it? Do we know that this isn't just the product of some computer time that it really applies to the real world?
Cristina - Yes. We have data from existing wind farms. So, I can definitely access those and that's in my list of things to do. The tools I'm using right now, the simulators are more useful when you're planning for a future farm. You don't have data because it's a prediction. And so, I'm trying to tune my model so that it works very well with existing farms, and so that I can use it now and simulate conditions in other locations for future times and so on.
15:48 - Maths gives cancer clues
Maths gives cancer clues
Fresh insights into the development of cancers has been revealed by a new study.
The story starts more than 100 years ago, when biologists looking at cancer cells down the  microscope noticed that the chromosomes - the long strings of DNA - in cancer cells were unusual, with cells often having the wrong number of them. Healthy human cells have 23 pairs of chromosomes, but cancer cells may be missing one of some pairs - a phenomenon known as aneuploidy - and have extra chromosomes in other pairs. There can also be smaller-scale faults, such as large chunks of chromosomes being copied or missing.
microscope noticed that the chromosomes - the long strings of DNA - in cancer cells were unusual, with cells often having the wrong number of them. Healthy human cells have 23 pairs of chromosomes, but cancer cells may be missing one of some pairs - a phenomenon known as aneuploidy - and have extra chromosomes in other pairs. There can also be smaller-scale faults, such as large chunks of chromosomes being copied or missing.
But it's not been clear whether this is just an effect of all the other things that are going wrong in cancer cells, or whether it's a key driver of the disease. And until now, most cancer genetics researchers have been focusing on smaller faults in genes, known as mutations, rather than such big changes. But there's growing evidence that there may be some kind of pattern to the chromosome changes in cancer cells, and understanding them could be the key to developing more effective strategies for targeting the disease.
Rather than doing experiments on cancer cells in the lab, the Harvard team have used mathematical analysis to look at the chromosome changes in the DNA sequences from more than 8,000 cancer samples stored in three gene databases. In particular, they were looking at genes that drive cancer cell growth - known as oncogenes - which are often overactive in cancer, and tumour suppressors, which act as the cell's "brakes", which are often missing or faulty.
Publishing in Cell, they found that regions of chromosomes harbouring many tumour suppressors were more likely to be missing, while areas packed with oncogenes tended to be copied. And regions containing particularly potent oncogenes or tumour suppressors were more likely to be copied or missing, respectively. This suggests that these chromosome problems are a key driver of cancer, rather than just a consequence.
The study also questions the classic "two hit" hypothesis of cancer development, which says that you need to have faults in each of the two copies of a cancer gene in the cell to promote tumour growth. But the team's results suggest that adding up the effects of losing several tumour suppressors in a row from just one chromosome, or copying several oncogenes, can be big.
For now, this idea is still theoretical, as the researchers need to go on and do experiments with cancer cells in the lab to prove their findings are correct, adding or removing crucial regions of chromosomes and seeing if they drive cancer. And it's not clear how this new mechanism fits in with other things that we know are important in driving cancer, such as faults in individual genes, or changes in their activity levels. But, for now, it's certainly a fascinating new idea to be explored.

18:56 - IBM vision for computer of the future
IBM vision for computer of the future
International computer giant IBM has unveiled its vision for a "supercomputer in a sugarcube"  powered by "electronic blood".
powered by "electronic blood".
The aim is to base the design on the workings of the human brain in order to surmount some of the difficulties - delivering power and removing the excess heat - that computer engineers are currently encountering as they seek to make chips smaller and faster.
The heat problem effectively doubles the running cost of a computer, because nearly as much energy has to be spent on cooling as on computing.
Current figures suggest that just running the Internet accounts annually - in energy terms - for an equivalent amount of CO2 as the airline industry, and computing generally equates to a global spend of over $30 billion just making hot air.
But, to make processors more compact and run even faster, chips will need to shrink, partly to reduce the distances - and hence time - over which information needs to be transmitted and to pack in more processing power. Doing so, though, will intensify the present problems.
So IBM have turned to the human brain for inspiration because, despite consuming energy at the rate of only about 20 watts, the brain is, according to IBM researchers Patrick Ruch and Bruno Michel, "10,000 more dense and efficient than any computer around today."
This they put down to the fact that the brain solves several problems in one: the 3D structure of the brain makes it highly compact, and it uses a highly efficient circulatory (blood) system to deliver energy and keep the operating environment right for optimal performance of the components.
According to Michel, the brain uses "40% of its volume for function and 10% for energy and cooling." The best current computers, on the other hand, devote only about 1% to processing!
Instead, Michel and Ruch propose to pile up processors like the chip equivalent of a skyscraper to boost computing power; then, they'll keep them cool by circulating a fluid - "electronic blood" - through special channels within the chips to soak up heat; and - here's the really clever bit - they'll also use the same fluid to power the chips using a chemical reaction.
Analogous to blood delivering sugar to hungry neurones, the approach being explored at IBM is to use a solution of a chemical like vanadium that can be "charged" before it is pumped into the computer.
Then, as it passes over the processor components, the vanadium alters its chemical oxidation state, releasing electrons to the transistors to power them.
The ultimate goal is to cram a super-computer that currently occupies half a football pitch into a volume the size of a sugarcube by 2060.

21:19 - What is Cancer?
What is Cancer?
with Gerard Evan, University of Cambridge
The National Cancer Research Institute's annual conference took place this  week, bringing together the whole cancer research community in the UK and beyond, from lab researchers, doctors and nurses to patient groups and research charities. Kat Arney was joined by Professor Gerard Evan from Cambridge University, who opened the conference with a fantastic talk about his work towards developing new approaches for treating cancers by finding common targets across many types of tumour.
week, bringing together the whole cancer research community in the UK and beyond, from lab researchers, doctors and nurses to patient groups and research charities. Kat Arney was joined by Professor Gerard Evan from Cambridge University, who opened the conference with a fantastic talk about his work towards developing new approaches for treating cancers by finding common targets across many types of tumour.
Kat - We hear a lot about the term 'cancer' and also its different types - breast cancer, lung cancer. But what is cancer?
Gerard - Well, different cancers are different. But by and large, tissues in our body behave themselves. They stay the same size and if we damage them, they regenerate, and they re-grow, and they go back to the same size. So, they're very tightly controlled. Cancers are cells that have lost control. They don't die when they should. They don't stop when they should. They grow, they spread, they invade, and they kill.
Kat - And what's actually driving these cells to go wrong because they start in our body? You'd think, what's making them go rogue.
Gerard - Yeah, well this is why cancer is so interesting, because for things to go wrong and cancers to have deregulation of their growth and their survival, that implies that normally, these processes are very tightly regulated. And so in a sense, understanding cancer is to understand how normal processes of the normal body is maintained in the beautiful way that it is for so many decades. We know that there are genes which make proteins, and these proteins act as machines that move information around. And they get information from outside and they transfer it to the various places within the cell that then execute that information. And some of that information make cells proliferate, grow; some of it makes cells stay alive; some of it makes cells differentiate into other cell types and move around. And when the genes that make these proteins, these engines that communicate this information, make defective versions, sometimes these engines can work without a signal. And so, they send a go signal all the time or stay alive signal all the time. The cells don't stop and the cells don't die.
Kat - So, we've got these genes going wrong in our cells and making them grow out of control, but we have lots of different types of cancer. We have breast cancer and lung cancer. How different at this kind of molecular level are these different cancers?
Gerard - Well, we don't have that many genes. We've got about 20,000 genes. So, there's a tool kit that's used again and again and again, but in slightly different variations. So the basic machinery of the cell cycle - how a cell replicates itself is pretty much the same in every cell type. What differs is the signals that trigger that decision of the cell to replicate itself and the various mechanisms that stop that happening. And they will vary from tissue to tissue. Tissues have different architectures, they have different risks of infection, different risks of these mutations arising. And so, it varies from cell type to cell type and tissue type to tissue type in a way that don't really yet understand.
Kat - But we do know a lot of things about how the cell cycle works, how cells multiply, but it still seems that although we have made progress in treating cancer particularly in recent decades, there's still a real challenge here and it does kill hundreds of thousands of people. Why is it so difficult to treat?
Gerard - Part of the problem is, that we know lots of the bits that go wrong, but we don't know how they fit together. It's a bit like saying, you've got an orchestra and the violins don't work very well, so that doesn't sound very good. But then we would fit all the instruments together and they were all working, and we'd still wonder why it was that we couldn't play a symphony. And that's because we're missing a bit which is the conductor. So the conductor is a different type of entity to the individual components. We're only just beginning to understand how the conductors work in this system. How, if you like, there's oversight over how everything integrates together. It's a much more complicated problem than looking at the individual bits.
Kat - The work you're doing is actually studying one of these conductors, a gene called Myc, which we know is important in cancer, but trying to understand what it does. How are you trying to use Myc to find better ways of treating cancer?
Gerard - The idea is - I mean, we know that cancers, they arise spontaneously within cells in our body and they evolve. So, everybody's cancer is different to everybody else's cancer. No two of the same and actually, lots of cells within each cancer are different because they're all evolving in different directions. But there's a great deal of commonality because of this common tool kit that iss used time and time again in different cells and different tissues. So, one of the ideas is, instead of looking at everything that's different from cancer to cancer, you instead focus on what's the same. This protein Myc is like a conductor. It doesn't regulate individual genes and how they're expressed. It seems to coordinate them altogether. And so, it acts as a central regulator. And so, the idea is if you hit that central regulator, maybe you'll hit many different types of cancer, maybe all types of cancer with just one type of drug. It sounds like science fiction, but we certainly think there's evidence to believe it's true.
Kat - And I saw your talk this afternoon and it was really exciting - some of the results that you talked about. What is your hope for the future? Do you think this could be a magic bullet?
Gerard - I don't know about magic bullets. I certainly think it's important to demystify cancer. There are lots of ideas that you can't cure cancer because it's endlessly adaptable and there's nothing we're ever going to be able to do about to really lick it. I don't believe that's true. Cancers at their root are relatively simple types of disease pathology. I just think if we think about them the right way, there's a good chance that we will be able to find therapies that are generally applicable against many different cancers. I'm very, very optimistic.
Kat - Thanks very much. That's Gerard Evan from Cambridge University.
28:07 - Screening Blood for Cancer
Screening Blood for Cancer
with Tim Forshew, Cancer Research UK Cambridge Institute
Historically, cancer scientists have studied tumours by taking samples from  them and then looking at them under the microscope, or they've used scans to pinpoint where cancers are. The problem with this is that to do either of these things requires the cancer to be of a sufficient detectable size. By the time it is detectable, often, it's usually very advanced. But now, researchers at Cancer Research UK's Cambridge Institute are pioneering a way to track tumours using fragments of their DNA that they release into the bloodstream. This means that the behaviour and the response of the cancers to treatment and even tiny tumours can be followed up. Chris Smith went along to see Tim Forshew, one of the scientists behind this new technique, to see how it works.
them and then looking at them under the microscope, or they've used scans to pinpoint where cancers are. The problem with this is that to do either of these things requires the cancer to be of a sufficient detectable size. By the time it is detectable, often, it's usually very advanced. But now, researchers at Cancer Research UK's Cambridge Institute are pioneering a way to track tumours using fragments of their DNA that they release into the bloodstream. This means that the behaviour and the response of the cancers to treatment and even tiny tumours can be followed up. Chris Smith went along to see Tim Forshew, one of the scientists behind this new technique, to see how it works.
Tim - It's been known for a long time that if you take a blood sample and you centrifuge it to get rid of the cells, you get what's known as blood plasma. And if you look in this blood plasma, you'll find copies of your genome floating around cell free. So, this is you're instruction manual. It's equally been known that at least in certain cancer patients, you'll find higher levels of this DNA floating around, cell free. And so, it's been hoped that you could use this as a way of detecting or monitoring and analysing cancer.
Chris - Is that genetic material intact? Could you read it like a book with all of the pages in sequence, or is it broken up like someone has shaken the book and all the pages have come adrift?
Tim - It's not intact and that's been one of the biggest challenges. So, these instructions are typically something in the region of 150 to 160 letters in length. So, we've got very small fragmented bits of DNA and very few copies of this DNA floating around in the blood.
Chris - Roughly, how much is in there?
Tim - So, in healthy individual, you'll have somewhere in the region of 2,000 copies of your instruction manual floating around in 1 millilitre of blood plasma. In someone that's got cancer, you may end up with one or two copies of your cancer instruction floating around or may have many copies of your cancer instructions floating around.
Chris - How are you going to read then all these mixed up DNA which is basically 160 letters long out 3 billion in the average person and put it back together? So, you're putting all the pages back in the book in the right sequence.
Tim - So, for some time now, people have had methods where they can look for one particular fault that is known to cause cancer or maybe a couple of faults. But the focus of our research has been to see if we can actually, as you say, read these instructions and look at large regions of the genome and try and understand what is going wrong in cancer and see if we can detect or monitor cancer through looking in the blood. So, we've been doing this with an approach called Next Generation Sequencing. If you come through here, then I can show you the machines we've been using.
So, this is one of our Next Generation Sequencing machines, and the way that we use this is we first take a blood plasma sample and we extract the DNA from this. Then we use a process called the polymerase chain reaction, or PCR, and this is a process that allows you to copy just certain parts of your instruction manual. So, to do this, you design something called primers which are short bits of DNA which you target to the specific - in our case, cancer - gene that we would like to read. You use these to pick out and amplify just certain parts of the instruction manual that you're keen to analyse. Then what you do is you take these amplified copies of your instruction manual and you put it on to one of these next generation sequencing machines. This then allows you to go through and read letter by letter all of these instructions and try and find where faults may lie, where cancer-specific changes are.
Chris - So, some of the genes that are in the person's cancers have got genetic spelling mistakes, for want of a better phrase, and that makes the gene misfunction - go wrong - and that contributes to them having cancer or to the way their cancer behaves.
Tim - Exactly, right. So, cancer is essentially a fault with your instruction manual and it's where you get faults that give an advantage to a cell. This cell will then grow more than it should. By finding these faulty instructions, we have a way of looking in blood and understanding what's going wrong in the cancer and potentially, working out how big the cancer is, or how well your treatment is working, by quantifying how much of these faulty instructions are floating around.
Chris - And so equally, if someone has a certain gene that's gone wrong that leads to their cancer not responding to a certain drug, you're going to see that and that means you can predict that happening before the person even realizes it, and so, you could change the drug treatment for example.
Tim - This is certainly what we're hoping. So, we need to do more research in understanding different types of cancer, how much of this DNA is being released. But certainly, from the early results that we and other groups around the world are gettingg, it suggests that at least in certain types of cancer, you will, as you say, be able to understand what's going wrong in the cancer before you can even necessarily see it through other processes. And then start to tailor the way that you treat an individual patient.
Chris - And monitor their response going forward.
Tim - Absolutely. So, once you found these particular genetic changes that are floating around in the blood, we hope that, again, for at least certain types of cancer, you'll be able to see if a mutation is decreasing and your treatment is being successful, or if the levels are increasing and your treatment should potentially be changed. And indeed, whether new mutations, new faulty instructions are appearing, suggesting that the cancer has evolved and you now need a different treatment.
Chris - I suppose this must give you as a scientists a totally new insight into cancer though because where previously someone might have been able to give you a little bit of one part of one cancer from one person. Now, if you take just a blood sample, which is going to be better for the patient, you're going to get a cross section of potentially all of the tumours including those tumours that have spread around their body all at once.
Tim - Absolutely. So, the challenge is, how much of this DNA is released and this is still something we're assessing. But assuming sufficient quantities are released, then as you say, it allows you to profile different parts of the tumour and equally, throughout the course of an individual's treatment. So, where getting a biopsy is often not even possible, with blood, you can take blood samples very regularly and start to assess how a tumour is changing.
Chris - It's almost like a liquid biopsy.
Tim - Absolutely. So, this is the term that we're starting to use for it.
35:54 - Cancer Screening Tools
Cancer Screening Tools
with Nick Coleman, Professor of Molecular Pathology, University of Cambridge.
Improvements in monitoring tumours can help us improve cancer treatments, but what about diagnosis? Biological markers might offer us a way to detect cancer in a sample more quickly than scanning thousands of cells to look for changes that might tell us that they're bad. To find out more, Kat Arney was joined by Nick Coleman, who is Professor of Molecular Pathology at the University of Cambridge...
Kat - What does a good screening test for cancer look like?
Nick - Well, it needs to be accurate, really, so that if a patient has a cancer the test needs to be positive, and if a subject doesn't have cancer then the test needs to be negative. That balance between avoiding false positives, avoiding false negatives is the key to an effective test. Obviously, it also needs to be acceptable to the patient. You don't want to be doing something that the patient wouldn't want. It needs to be affordable. There are practical issues as well, but I would say accuracy is the key.
Kat - And it sounds such a simple thing although I know it's not - it needs to be accurate and only spot cancers and not spot non-cancers. But what are the problems with some of the current tests we have? What are some examples of the current screening tests that don't really answer those questions?
Nick - So, bowel cancer screening is one example, where at the moment, people are being asked to provide a stool sample which is then tested for the presence or absence of blood. And that test is better than nothing, but it's far from perfect because there are patients with the cancer who do not have blood in their stool and there are patients without cancer who do. So, we end up with fairly limited accuracy. The requirement for lots of patients who don't have bowel cancer is to undergo some pretty invasive second stage investigation to see whether or not they generally have a cancer or not. So, there's scope for improvement in several of the cancer screening tests that we currently have.
Kat - Here in the UK, we currently have national screening programmes for breast, cervical and bowel cancer. Could we screen for all cancers and why don't we?
Nick - It would be nice to be able to do that, but I think we have to be realistic about the strengths and weaknesses and what currently can be done. One example is prostate cancer where it would be marvellous to be able to detect the cancers earlier and treat the cancers that need to be treated. But the test that is currently available is a blood-based test for a protein that's made by prostate cells. But again, it lacks the ability to detect cancer in all patients and in particular, it is prone to false positive results so that patients without cancer in their prostate have a positive result or patients who have a cancer, are identified, but those cancers are not destined to do any harm to the patient. This is a problem of so-called 'over diagnosis'. That's a real issue in prostate cancer, and it's currently estimated that something like 50 men have to undergo cancer treatment to save one life from prostate cancer. So, There's a real opportunity and need for new screening approaches in prostate cancer and others too.
Kat - So, tell me a bit about your work. What are you trying to do to address these needs?
Nick - Well, we work in cancer of the cervix or screening for prevention of cancer of the cervix, where again, this is a test which has value of course, but also has opportunities for improvement because an individual cervical smear test - if a woman has a pre-cancer in the cervix - an individual smear test is going to be positive in only about 50 to 70% of cases. The reason for that is that currently, it's a human activity which involves a subjective assessment. So, decision making by individuals, trying to look for very subtle differences between a pre-cancer cell in the cervix and a normal one. So, what we've been trying to do is identify biological differences between a cancer cell and a normal cell in the cervix, so that we can stain pre-cancer cells a different colour from their normal counterparts. And by being able to do that, it makes it much easier for these pre-cancer cells to be seen. It means you can do it more quickly, more accurately, more cost-effectively. There are multiple practical benefits in being able to do that. And so, the technique that we've developed is currently undergoing trials. We're very encouraged by the results of them so far and we're hopeful that there will be a contribution at least from this test to cervical screening.

40:43 - Making Cancer Visible to MRI Scanners
Making Cancer Visible to MRI Scanners
with Kevin Brindle, Cambridge University
Kevin Brindle, from the University of Cambridge, has developed a way to trace the location, the growth and the activity of cancers inside the body. With a very powerful magnet, he makes the nuclei at the centres of carbon atoms, in things like sugars, all spin in the same direction. Injected into a patient, these sugars are taken up and used by tumours where the magnetically aligned spins of their carbon atoms gives them away to a magnetic resonance - MRI - scanner, so it can tell where in the body the tumours are. Ginny Smith went to see how it works.
Kevin - Conventionally, the way we assess treatment response in cancer is to look after drug treatment, whether the tumour is shrinking. The problem with that approach is it can take many days, weeks, or even months before you see any evidence of the patient responding to treatment. What we've been interested in doing is imaging tumour biology, which can give a much earlier indication of whether a drug is actually affecting the tumour and whether the patient therefore is responding to treatment or not. With modern therapies, what we know already is that not all patients respond in the same way. And so, when you give a patient a new drug, you want to find out very quickly whether that drug is working. With the techniques that we're developing, we think we can tell within hours or certainly days whether anything is happening. Clearly, if the drugs not working in that particular patient then you can try an alternative approach.
look after drug treatment, whether the tumour is shrinking. The problem with that approach is it can take many days, weeks, or even months before you see any evidence of the patient responding to treatment. What we've been interested in doing is imaging tumour biology, which can give a much earlier indication of whether a drug is actually affecting the tumour and whether the patient therefore is responding to treatment or not. With modern therapies, what we know already is that not all patients respond in the same way. And so, when you give a patient a new drug, you want to find out very quickly whether that drug is working. With the techniques that we're developing, we think we can tell within hours or certainly days whether anything is happening. Clearly, if the drugs not working in that particular patient then you can try an alternative approach.
Ginny - So, what kind of things can you look at within a tumour to see if it's being affected by a drug?
Kevin - In our work, what we do is, we take a carbon-13-labelled molecule. We can inject that into the patient and then we can image where that carbon-13-labelled molecule goes. We can image it being used by tumours and, frequently, when we treat a tumour, we see a decrease in the rate at which they use this molecule.
Ginny - So, do you put this molecule within a larger molecule?
Kevin - We have recently used glucose but we've mainly used the molecule called pyruvate, which tumours rapidly take up and convert into another molecule called lactate. And how fast they do this tells us something about how healthy that tumour cell is.
Ginny - Now, I think you've got an example of a machine that actually creates that sample that you can inject. Can I have a look at it?
Kevin - Sure. Shall we go down and look it in there?
Ginny - Yes, sounds good. Okay, so we're walking into - well, it doesn't quite look like a normal lab. It looks more like maybe a boiler room. So, there's two big metal, shiny containers and lots of wires and hoses and things coming out in various machines with lights on. Can you talk me through what some of the intriguing looking tubes and knobs and things on it are?
Kevin - Yeah. So, what we're looking at here, this can is a magnet. It operates 3.35 tesla and to put that in context, the Earth's magnetic field is about 50 micro tesla. So, we're more than a million times stronger than the Earth's magnetic field.
Ginny - That would confuse anyone around here with a compass.
Kevin - It certainly would. Okay, so what we do is we put the sample into the bore of this magnet and then we bleed liquid helium on to the sample. So, liquid helium is about 4 degrees Kelvin minus 269 degrees centigrade. And then we put on a vacuum and that noise you can hear is the vacuum pumps running. So, by boiling off the helium, the energy that is used in that takes the temperature further down to 1.2 degrees Kelvin. And at that temperature, the spins become polarised.
Ginny - So, what exactly do you mean by that?
Kevin - For example, the spins that we're interested in - carbon-13, which is an isotope of carbon - if you put those nuclei in the magnetic field, they behave like a bar magnet. Some of them line up with the field and some of them line up against the field. You have a slight excess that line up with field. And it's that polarisation, that slight excess, which gives rise to the signal in the MRI experiment. Okay, now we have a problem. We have our sample where we polarise all these spins, but it's at 1 degree Kelvin. The key now is we need to get it out, very, very fast, and if we can do that, we can retain the spin polarisation. And that's what this device here does.
Ginny - Okay, so that's sort of a long tube with what looks like some kind of gauge at the top and another tube coming off it that looks like you might be able to connect it to something.
Kevin - That's right.
Ginny - What does that do?
Kevin - Okay, so we put what we call a dissolution fluid into here. We heat it up under pressure. Temperature goes up to about maybe 200 degrees centigrade. We then open this valve here that blasts this super heated water on to our frozen sample and blasts it out of the magnet very, very quickly, so fast that we retain the spin polarisation. So, these nucleus spins, these carbon-13 spins that are all lined up now with the field, we keep that, but we lose it quite quickly. As soon as you bring it up to room temperature, you start to lose this polarisation. So then we run over this other magnet and in this magnet, we'll have the system that we want to image. We can then inject this material and now, we have so much signal that we can image where this sample goes in the body and we can then watch how it's used by cells in the body, how it's metabolised, how it's converted from one molecule into another. We can watch that in real time.
Ginny - So, this doesn't look like the kind of MRI machine I've seen in hospitals and things. I'm assuming you don't put a human in this one.
Kevin - No, it would have to be a fairly slim human! The bore size of this is about 9 centimetres, but we have recently acquired a clinical device, which is sat next to a clinical machine up in Addenbrookes Hospital in the Department of Radiology. In a year or so's time, we hope to be doing exactly this experiment that I've just described here, in patients.
Ginny - Now, these machines, they're quite big and bulky. Is that exactly what you'd have to have in a hospital?
Kevin - The clinical device actually is fairly compact, standalone. It will stand just outside the room where the imaging magnet is, and it will spit out labelled material, polarised, that we can inject straight into the patient.
Ginny - How long do you think it'll be before we're seeing this as standard in all our hospitals?
Kevin - We hope that it will be in widespread use in 10 to 15 years.
Screening Cancer: Scanners and Antibodies?
Nick Coleman - So, the first thing to say is, that wouldn't be screening. Screening is testing for the presence or absence of disease - in this case, cancer - in somebody who's symptom free. But in terms of a diagnostic workup of somebody who has symptoms or has a known tumour, this kind of approach I think is very exciting because as I was saying earlier, as we now understand more about the biology of tumours, we can pinpoint differences between tumour cells and normal cells that will allow us to target their presence. One example that's being looked at quite a lot is molecules that are secreted by cancer cells which provide them with more blood supply. Because we know that the tumour cells are rich in these molecules, antibodies targeted against those molecules and then used in some sort of imaging process will allow us to pick up the presence of tumours or tumour deposits very accurately with a greater sensitivity than the existing imaging methods.
Using Viruses to Engage and Destroy Cancer?
Kat - Well, this is certainly a really exciting area of therapy. It's called "oncolytic viruses", kind of meaning, cancer bursting viruses. There's a lot of different trials underway. There's lots of different types of viruses. Adenoviruses are one of the types of viruses that people are using because they infect a lot of cells. The idea is that because cancer cells, as Gerard said, are kind of genetically compromised, they've got things wrong with them, they're not working the right way. So, you send in a virus that basically takes advantage of that, that can only replicate in cancer cells, and then it replicates and it bursts the cancer cells and they produce more viruses as they burst and they infect more tumour cells. But the idea is, they wouldn't infect healthy cells so you'd only be killing the cancer. That sounds like an absolutely fantastic idea and in the lab, these work really well. The trouble is, when you try and put them into trials in patients and sort of scale up the system, they stop working so well and one of the big problems is that the immune system kicks in, recognises the virus and mops it all up. So, there's some interesting work going on about how you could maybe suppress the immune system so you could get the viral infection in and then kind of let the immune system go again. That's being investigated in terms of gene therapy, which works in a similar way - you're trying to infect specific cells to deliver a payload. So, it's a really exciting area of research, but not quite paying off yet.
Chris - I went to a conference recently where, actually, they were saying that that immune response can be quite valuable because if you get the virus going into the tumour, it then drives the immune system nuts because the immune system sees this cancer growing loads and loads of virus. So, it reacts to the virus, but in the process, also reacts to the cancer and then the same immune response goes elsewhere in the body and starts attacking any spread of the cancer elsewhere in the body. So, they think this may actually be quite constructive.

50:21 - Can Superfoods Help Prevent Cancer?
Can Superfoods Help Prevent Cancer?
Hannah - So, we often hear headlines that foods like red wine and veggie curry might decrease your likelihood of cancer. Dr. Emma Smith at Cancer Research UK has this to say...
Emma - We hear an awful lot in the newspapers and on TV about these so-called superfoods or groups of food that allegedly kill cancer. In fact, there's just isn't any solid evidence for this at all from clinical trials. There isn't any single food, or type of food, that will protect you from cancer ultimately. These stories normally come from research carried out in the lab where chemicals purified from food or perhaps just known to be found in certain foods, like broccoli or cabbage, are shown to kill cancer cells when the chemical is put on the cancer cells grown in a dish in the laboratory.
Hannah - Okay, but surely, this lab data somehow translates to how my body reacts to particular foods.
Emma - Now, the problem is, we don't necessary get those chemicals in that form from food or perhaps we don't get the chemicals in the kind of doses that they're adding to the cancer cells. When you look at the evidence from clinical trials, there's just not been any proof that these foods help protect anybody from cancer.
Hannah - And what about red wine? I'm sure I've heard that that's a good cancer beater or is that just me being a positive glass half full kind of girl?
Emma - Well, red wine is an interesting one and we are actually running clinical trials in Leicester looking at - it's the chemical in red wine called resveratrol and there's also, perhaps you've heard about curcumin and turmeric which is the colouring they put in curry. Now, there is some evidence that those chemicals could protect us from cancer. So, we're running a clinical trial to look at whether this is the case or not. But it's important that we're actually studying the chemicals in a purified form in controlled doses. We are not getting people to drink red wine and eat curry. In fact, the evidence is, that the alcohol in red wine far, far outweighs any benefit you would possibly ever get from resveratrol. So, we definitely are not cordoning red wine to protect you against cancer.
Hannah - And a paper published earlier this year in the International Journal of Cancer used clever chemical analysis. And it turns out that 111 glasses of red wine need to be consumed each day in order to get the correct dose of cancer beating chemicals to balance out the carcinogenic ethanol, which is a bit excessive an intake, even for me. So, is there anything that somebody with cancer can do whilst undergoing treatment? Can they help by changing their diet?
Emma - First of all, if you're undergoing treatment, treatment is quite challenging for the body. So, it's important that you actually make sure the calorie intake is high enough so you can cope with the treatment. If you are considering any major changes in your diet, whether it's to lose a bit of weight or just eat healthier, it really is important that you consult your doctor first before making any major changes.
Hannah - Thanks to all those listeners including Calind and Kate who got in touch with the question and also to Dr. Emma Smith.
- Previous Tamoxifen: Chemistry in its element
- Next Bodyguard drugs and TB










Comments
Add a comment