What are legal highs, and how do scientists, doctors and law-makers keep up with new drugs entering the market? Plus, biofuels and why they cost the Earth, the cause of LED droop, a neutron star proves Einstein's theory of general relativity right, and E. coli programmed to pump out diesel.
In this episode

01:03 - The Trouble with Biofuels
The Trouble with Biofuels
Biofuels - often suggested as low or no carbon alternatives to fossil - may not be as green as they seem.
The promise is attractive: can we use plants to make fuel? If we could, we would be taking carbon dioxide out of the atmosphere and using sunlight to turn it into fuel. The carbon dioxide would of course end up back in the atmosphere after we burned it, but that's got to be better than burning fossil fuels and turning trapped carbon into nasty CO2. Hasn't it?
Unfortunately, it probably isn't.
In 2013-2014, biofuels will make up 5% of the UK's transportation fuel, either as bioethanol in petrol or as biodiesel. By 2020, this could be as high as 10 or 15%. The bioethanol is produced either from corn (maize) grown in the US or from Brazilian sugarcane. The biodiesel is mainly from waste cooking oil or other non-food sources such as tallow - animal fat.
The problems with using potenti al food sources to produce fuel are relatively well known - food prices are increasing in the developed world while much of the developing world is struggling to get enough to eat, so diverting crops to produce fuel seems counter-productive. In the case of US corn, for example, using the crop to produce fuel means that the price of corn to eat goes up. So what, you might ask, I can live without sweetcorn? But corn is widely used as an animal feed, so the price of beef goes up. Livestock farmers then look for alternative feed and the price of other grains - wheat, barley or rice - goes up. So using corn for fuel makes the meat and the bread in your burger more expensive!
al food sources to produce fuel are relatively well known - food prices are increasing in the developed world while much of the developing world is struggling to get enough to eat, so diverting crops to produce fuel seems counter-productive. In the case of US corn, for example, using the crop to produce fuel means that the price of corn to eat goes up. So what, you might ask, I can live without sweetcorn? But corn is widely used as an animal feed, so the price of beef goes up. Livestock farmers then look for alternative feed and the price of other grains - wheat, barley or rice - goes up. So using corn for fuel makes the meat and the bread in your burger more expensive!
Of course, pricier burgers in the developed world might actually be a good thing with our obesity epidemic, but in the developing world higher food prices can be catastrophic. Food riots and political instability can result.
So those are the questions about social/political sustainability that biofuels raise, but what about the environment?
In order to produce biofuels from crops without affecting the global food supply, we'll need to use land not currently used for farming. And the big problem with that is that it releases a lot of carbon that is "locked" in either forests or peatlands. Palm oil production is linked to destruction of rainforest in the tropics, for example, so there are losses in biodiversity as well as in "locked" carbon.
So even if the resulting biofuel is produced from then on in a low-carbon process, the net effect is still contributing to global carbon emissions.
What about biodiesel waste products? Although these are much better, there is still some uncertainty about exactly how much better. And some estimates even suggest that using them might increase overall carbon emissions - for example, if tallow is used for fuel instead making soap or animal feed, it could be replaced with something derived from fossil fuels. And in the case of used cooking oils, it's very hard to trace the product - it could be that palm oil, produced in an area that was once rainforest, is sold as recycled when it hasn't been used. That might sound like a strange thing to do, but if the price of palm oil compared to used cooking oil is right, then it's worth doing - especially when recycled oil counts double towards a company's biofuel credits compared to 'fresh'.
And finally, all of this is expensive - estimates start at $200 per tonne of carbon saved and go up from there. These costs will be borne by motorists (10 cents per litre - but that might be for a whole litre, rather than the 5% mix). Chatham House estimate the cost at around £460 million a year - around £30 to £40 per motorist. They suggest that there are better ways to reduce carbon emission from transportation that actually save money in the long run - switching to more efficient cars, for example. And in the longer term, hydrogen, battery or fuel cells will be a better way to remove carbon from cars.

04:45 - Reducing the Droop in LED Efficiency
Reducing the Droop in LED Efficiency
A team from the University of California, Santa Barbara (UCSB)  have answered an important question in LED performance - called 'efficiency droop' - which has limited the development of high-performance LEDs, capable of replacing incandescent and fluorescent bulbs.
have answered an important question in LED performance - called 'efficiency droop' - which has limited the development of high-performance LEDs, capable of replacing incandescent and fluorescent bulbs.
Light Emitting Diodes (LEDs) are highly efficient, durable, and long lasting, and they've found uses in everything from remote controls to traffic lights. Unlike incandescent and fluorescent bulbs, they don't have a filament that will burn out, they don't get particularly hot, and with a lifetime of tens of thousands of hours, they greatly surpass the performance of an incandescent bulb.
But LEDs have traditionally suffered from an effect called 'efficiency droop', a drop in the light output of an LED that occurs when a high current is applied to the device. This has meant that an LED's "cost per lumen" - a metric that reigns supreme in the lighting world - is higher than that of the more traditional large-scale lighting solutions, and this is holding back the more wide scale implementation of this novel light source.
Now this may be about to change. In 2011, a team led by Chis van de Walle at UCSB, theorised that behind this droop was a complex non-radiative process called Auger recombination. When this happens, the charge carriers in the LED material, known as electrons and holes, lose energy as heat rather than light. Now a team led by another UCSB scientist, Claude Weisbuch, has proved the theory by analysing the electron emission spectrum of Indium Gallium Nitride (InGaN) LEDs.
In their paper, in Physical Review Letters, they study an LED made using thin layers of GaN-based material grown on a sapphire surface. By measuring the LED's light intensity, while directly observing the energy spectrum of electrons emitted from the device, Weisbuch and his team confirmed that Auger recombination was the culprit of the efficiency droop. The charge-carriers (electrons or holes) were interacting without giving off light, fully accounting for the drop in LED efficiency at higher currents.
In LEDs, electrons and holes are continuously created. When they recombine into electron-hole pairs, they release energy in the form of light, making recombination fundamental to an LED's operation. In nitride-based LEDs, three- particle - or Auger - interactions can also occur. Here, although an electron-hole pair is created, the resulting energy is not released as light, but instead promotes an unstable electron into a higher energy level - i.e. it heats that electron up. As you increase the current applied to an LED, you increase the number of unstable high-energy electrons in your material, increasing the change of forming a three-particle interaction.
Although this recombination effect cannot be eliminated, it can be minimised by engineering new materials designed specifically to combat it. So this study is likely to lead to new LEDs with significantly higher light emission efficiencies.
The U.S. Department of Energy recently estimated that 12% of total U.S. electricity consumption is is used for lighting, so this development has far wider implications than just the academic. Higher-efficiency LEDs have enormous potential for use in long-lived, reliable lighting for residential and commercial applications.

09:24 - Proving Theories of Gravitation
Proving Theories of Gravitation
Observations of a massive neutron star in a compact stellar binary in outer space confirm general relativity theory in a previously untested regime, a new study reports.
General relativity (GR), proposed by Albert Einstein, has been tested outside of Earth before, but it has not been tested in the strong field of such a massive neutron star (or other, even stronger gravitational fields).
Scientists haven't known whether this theory applies in such extreme  environments. Now, new research by John Antoniadis and colleagues tests this theory in a strong-field setting.
environments. Now, new research by John Antoniadis and colleagues tests this theory in a strong-field setting.
Antoniadis and colleagues report the measurement of a massive neutron star twice as heavy as the sun; because of their extremely high densities (which imply strong gravity), neutron stars can be used to test gravity in a strong-field regime. The researchers identified this particular star using spectroscopy; it showed up as a millisecond pulsar. Critically, and unlike other massive neutron stars known today, it is in a very tight orbit, requiring less than three hours to circle around another star. This makes it possible to measure the orbital decay resulting from the gravitational waves it emits.
The researchers have been observing changes in this binary's orbital decay since 2011. Thus far, they find these changes to be consistent with the rate of decay predicted by general relativity. This observation rules out the strong-field phenomena predicted by alternative theories of gravity and provides support for the validity of Einstein's theory, even in the extreme conditions present in this system. The work of Antoniadis and colleagues may also support the use of GR-based templates for gravitational wave detection here on Earth.
Meanwhile, because the binary system in which this massive neutron star was observed has a peculiar combination of properties related to its mass now and what scientists believe such an old star's mass should be, it poses a challenge to the current understanding of stellar evolution and will likely motivate further study.
13:31 - Sounding Sexy
Sounding Sexy
Marilyn Monroe and Barry White had it sussed decades ahead  of the science. Marilyn's breathy vocals takes many men's breath away. And Barry's deep crooning has left millions of women swooning. But why is this?
of the science. Marilyn's breathy vocals takes many men's breath away. And Barry's deep crooning has left millions of women swooning. But why is this?
A paper published in Plos One this week by Yi Xu and colleagues at University College London revealed how you sound sends out signals about your body shape. Marilyn projects petite, while Barry sounds big and strong.
The vocal tract, or airspace above your larynx, shapes the resonance of the sounds produced by your vocal folds and is shaped differently in men and women, a trait thought to be under the control of sex hormones oestrogen and testosterone. These two hormones are also involved in body shape. So the sounds you make might also be linked to what your body looks like.
To test this, researchers took 16 males, 16 females averaging mid 20s in age, asked them to listen to the sentence "I owe you a yoyo," over 100 times.
The sentence was tweaked each time and volunteers had to rate the attractiveness of the sound.
Males rated females as more attractive if they had increased breathiness and dynamics of vowels (called wide formant dispersion). If the voices were made lower, they were less attractive. But increasing pitch didn't increase attractiveness. So a natural pitch seemed best.
Females, on the other hand, preferred a male voice that signalled a large body size via low pitch and monotone vowels. Interestingly women also seemed to prefer a breathy man though, which may soften the aggressiveness associated with large body size.
The evolutionary biology behind this? The vocal folds in our larynx knock repeatedly against one another as air rushes past from our lungs, causing vibrations and producing the voice. The frequency of the sound they produce is affected by their length, mass and tension. In men, the folds are longer and so produce sound of lower frequencies, which we hear as lower pitch; shorter folds in women will have a higher pitch. Females also have more breathiness which comes from air whistling through a gap at the back of the vocal cords. The vocal tract is also longer in men and it's this that makes their vowels more likely to sound monotone.
The scientist's hypothesise that there may also be an evolutionary pressure coming into play with vocal tract shape. Males across the bird and mammal kingdom use low monotone vocal sounds to project a threatening big body size to help successfully warn off predators and compete for a mate.
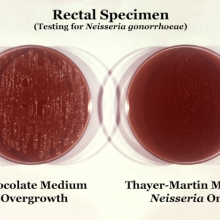
16:55 - Quickfire Science: Gonorrhoea
Quickfire Science: Gonorrhoea
This week, Professor Cathy Ison from Public Health England announced that the sexually transmitted infection gonorrhoea could become resistant to all known antibiotics by 2015. Here's the quickfire science on the disease from our Naked Scientists team members, Elena Teh and Pete Skidmore.
Elena - Gonorrhoea is a sexually transmitted bacterial infection most often seen in young adults in the age of 25.
Pete - With 62 million new cases each year worldwide, it's the second most common bacterial STI after chlamydia.
Elena - Gonorrhoea is on the rise in the UK with records showing a 25% increase in cases in 2011.
Pete - It's transmitted between people during unprotected sex and may also be passed from an infected mother to her baby during birth.
Elena - The bacteria can infect all parts of the female and male reproductive system as well as the eyes and throat.
Pete - The infection is often symptomless, but may cause yellow discharge and a burning sensation when urinating.
Elena - If left untreated, more serious complications can occur including the destruction of the Fallopian tubes and infertility in women.
Pete - Gonorrhoea is normally treated with a course of antibiotics, but some strains are developing resistance to these drugs.
Elena - Almost a quarter strains are now resistant to penicillin, so alternative antibiotics are having to be used. In time, these may also become ineffective.
Pete - With no new antibiotics in the pipeline, awareness of safe sex practices and regular check-ups is still the best form of protection.
18:44 - Running on bacteria: E. Coli & biofuels
Running on bacteria: E. Coli & biofuels
with Dr. John Love, University of Exeter
This week, a paper in PNAS revealed that E. coli bacteria can be used to produce biodiesel that is identical to the fossil fuel diesel that we currently use. This work has been pioneered by Dr. John Love. He's from Exeter University. John, why were you trying to do this? What was the motivation?
John - The original motivation was that, the current biofuels that we use in the first generation, ethanol and plant-derived biodiesel are actually not that good. Not just because of the problems that you were mentioning earlier but also because basically, in a car engine, they're not great, they don't burn very well. They have problems that clog up the engine, there are issues with quality control as well. And so really, what we wanted to do was to see if we can generate biologically, a fuel that was exactly what we needed for the retail fuel market. And rather than actually sort of having substitutes or additives, what we can do is have fuel replacements that wouldn't affect the efficiency of modern engines.
Chris - I was of the opinion that scientists are already modifying microorganisms to produce diesel and diesel-like substitutes - algae for example. So, what can you add by doing it in E. coli instead?
John - So basically, algae are very, very useful actually and I also do work a little bit on algae and I'm a big fan of algae. The problem is, is that the amount of land required to actually grow algae right now is substantial and also, the amount of capital expenditure into developing algal farms is very, very large right now. Even though you've got the concept of the integrated bio-refineries whereby you could use water treatment plants to feed algae and maybe nuclear power, hot water to actually heat them up, there are some issues regarding algae which you just can't get around and that is the amount of sun hitting the pond and the depth of pond, self-shading, and all these sort of issues.
Chris - And of course, E. coli will grow in the dark so you can get around that problem.
John - It will grow in the dark, it will grow in a great big tower so essentially, your land footprints are quite low and also the technologies of E. coli fermentation if you like are there in the pharmaceutical industry and the technologies of say, the brewing industry which relies on yeast can be fitted into an E. coli-style system.
Chris - So first off, what does it take to make E. coli make diesel because just in case anyone isn't aware of this, E. coli don't make diesel normally do they?
John - No, they don't, not at all. In fact, it's quite intriguing because a lot of microorganisms do make a lot of alkane-like compounds. A lot of animals actually make alkane-like compounds. For instance, birds use an alkane-like wax to waterproof their feathers. Plants use it on their cuticles to stop water evaporating from their leaves. Insects use it in their cuticles for the same reason. Cyanobacteria use it as well for a reason that we don't really understand just yet. But the thing is that E. coli does not do it. E. coli was a work horse of the laboratory and so, what we felt we'd do is, if we cannot find the organisms that can make the alkanes in sufficient amounts, can't we engineer them to make the alkanes that we need and that we want? So, what we did is we trawled the literature, trawled the gene bank and various things like that, came up with a few solutions that were possible and then built artificial synthetic metabolic pathways in order to generate the biofuels. And the conversion basically goes from free fatty acids to an aldehyde to the alkane. And we trim and we customise the chain length, the carbon chain length and the carbon chain configuration at the free fatty acid stage.
Chris - So in other words, what you feed the bugs determines what they churn out.
John - Well, not really, no, because actually, what we feed the bugs right now is glucose and really, glucose is a source of most of the energy in the E. coli cell and it will eventually be converted to a free fatty acid. If we were able to feed it another substrate, then it would just use it as an energy source and it would also take that to a free fatty acid, providing there's some carbon there, we can actually construct these fatty acid carbon chains.
Chris - So, you've taken chunks or modules of, I suppose, bits of metabolic pathways from a range of different microorganisms, reassemble them all in E. coli so it's got this ability to do this synthesis, it then makes these chemicals that to all intents and purposes are very similar in composition to diesel. How much can I get from one bacterium or if I grow a culture of them, can you get practical amounts out?
John - Well, right now, no, you can't and so, what we've done essentially, the proof of principle. What we need to do next is to actually work out the bottle necks in the metabolic pathways to try and iron out any competing reactions to make the process much more efficient, and potentially, also boost the amount of fatty acid that is produced by E. coli. And there's a wide literature in actually doing that. So, that might be relatively feasible. What we'd also like to do is alter the inputs if you like into the system, not just the outputs so that we can get the E. coli to feed off a variety of substrates and not just right now, glucose, but perhaps sugars derived from lignocellulose, so the degradation of wood or perhaps even waste products, we accept as useful right now.

25:05 - Il-Legal Highs: Telling the difference
Il-Legal Highs: Telling the difference
with Dr. John Ramsey, St. George's University of London
Hannah - Last year in the UK, 73 completely new legal high drug compounds were found in clubs and bought over the internet by the police. But how do scientists keep up with this constant flow of new and untested chemicals that are being released and flooding the market?
Chris - Analytical Toxicologist John Ramsey explained to me how his company, TICTAC Communications which produce a commercial drug database used by UK Healthcare and law and order professionals, track the appearance of new drugs, and what constitutes a legal high.
John R. - A legal high is a compound not controlled by Misuse of Drugs Act. Drugs that mimic existing drugs of misuse but are being modified sufficiently to just tweak them to bring them outside the Misuse of Drugs Act, but similar enough to maintain the pharmacology. Of course at the time they're synthesized and sold, nobody really knows whether they do that or not, but the assumption is that the molecular adjustments are relatively minor. Therefore, they will retain the pharmacology. Then they get marketed and evaluated by the users who report back whether they work or not and then they're either successful if they work as drugs or they're not successful, I guess, if they don't.
Chris - So, how do you stay on top of this?
John R. - It's a completely moving goalpost. We discover a new compound, we find out about it, we tell the legislators, they make it illegal, and then the bad guys look for a new compound. So, we go round and round this circle with a constant number of new compounds being made and detected, and then controlled.
Chris - So, do you have to dip into the marketplace - for want of a better word - to see what's doing the rounds then to keep updated?
John R. - Yes, we have lots of different strategies. I mean, we can only work within the law obviously, that's one issue. So, we use the contents of club amnesty bins to see what's being used in the great outdoors as it were.
Chris - Club amnesty bin?
John R. - Clubs are known to have a drug problem, so they need to do the best they can to minimise drug use on their premises. So, they will agree with the local police and the local licensing authority on a strategy to keep drugs out. And what it normally says is, they reserve the right to search people as a condition of entry to the club and if they find anything during the course of that search, as long as it's deemed to be small enough for people for personal use, as long as it's surrendered then people are allowed to go in the club, and the club operates normally.
Chris - And they dump the stuff into the bin and you're then able to access what they've dumped.
John R. - The police effectively collect the drugs from the amnesty bins and then they're brought to us by various police forces, then we analyse them chemically and then put them in our database.
Chris - How do you analyse a tablet you find in an amnesty bin?
John R. - The sort of routine methods. Essentially, infrared spectroscopy for powders because it's quick and cheap and then we follow that up with GC-MS, gas chromatography-mass spectrometry. The gas chromatography separates the drugs into its various components and the mass spectrometry effectively produces a fingerprint which identifies them.
Chris - How do you know this is a drug which actually will produce an effect in a user? In other words, it is a substance that has the potential to be illegal.
John R. - Well usually, they're sufficiently similar to an existing drug. I mean, some of them are literally minor modifications of existing drugs.
Chris - What does it take for legislation to get changed about a chemical? So, you find a chemical which has come out of a club amnesty bin or customs send to you and say we're finding this on people, does just the sheer fact that you find a new molecule immediately mean it gets regulated or is there a process there?
John R. - There's a process, but the problem is that in 2012, there were 73 new compounds notified to the European monitoring centre. Most of those compounds will be controlled on a European basis, but before that happens, they're risk assessed. So, just the fact that a compound exists doesn't necessarily mean that it will be controlled.
Chris - Where are all these new molecules coming from? Are there people sitting in their garden sheds across the country cooking up this stuff or is it more organised than that?
John R. - It's much more organised than that. They're almost all manufactured in China and imported into this country in bulk and then distributed. But whether the Chinese actually sit down and innovate the compounds and supply them or whether somebody in Europe or America commissions the synthesis, I think is unclear at the moment.
Chris - What about the fact that, yes, you could detect these things, you might find the one tablet in existence for that particular molecule, what about actually working out whether people are wholesale consuming an agent? How can we do that?
John R. - That's a big problem. We can lurk around the chat rooms and hear what people are talking about, but that's not terribly scientific, but it gives us an idea as to whether they work or not. But we also have a strategy where we analyse bulk urine samples and look for the presence of these things. Typically, urine collections from public urinals and we take samples from those and we can analyse them as an anonymous pool bulk sample and find out what's in them on a Saturday night in a particular district.
Chris - I do remember reading a paper maybe about 10 years ago or so where someone had gone and collected samples from the River Po in Europe and said there must be something like a million doses of cocaine washing down the river everyday based on their findings. So, you can just go and analyse river water to find out what people are using. Can you sort of go to that extreme with these other molecules?
John R. - We absolutely can. I mean, the pooled urine analysis has more sensitivity. You know, we're collecting undiluted urine from a couple hundred people typically. When you get on to sewage treatment works, the number of drug uses contributing to the pool has to be quite high before we can detect it. Nonetheless, we can easily detect MDMA from ecstasy tablets, cocaine, heroin, methadone, and whole variety of other compounds as well. And indeed we have a project called SEWPROF which is researching exactly these issues. We're looking for differences in drug use in different countries and in different populations. We're looking for the effectiveness of strategies to try and defeat drug use. And we're also looking for the occurrence of these new compounds as they become available.
Chris - When I was a medical student, we were shown a video of the very famous case of the MPTP story. Someone was trying to make a heroin substitute and lots of people turned up with the symptoms of Parkinson's disease and that always stuck in my mind and I thought, if I was ever offered a tablet in a club, I would never take it because the person who made that drug did not know they put these other toxin in by accident. Are you quite worried by what you see cropping up in the chemicals that you've analysed?
John R. - Absolutely. I mean, the circumstances you describe were before the internet. I mean, goodness knows what would've happened if somebody had made that now. At the time, it was distributed to only about 15 or 16 people locally. But now, when the stuff is made, it's distributed to hundreds of thousands of people potentially across the whole of Europe or even across the whole of the world. So, there is the potential for an absolute catastrophe and I think young people just don't realise the potential risks they're running. They don't understand why the pharmaceutical industries spent hundreds of billions of dollars and 5 years risk assessing stuff before it ever gets near a human volunteer. This stuff is completely untested and probably the first person who takes it is somebody who buys it either off the internet or even from a high street shop.
33:18 - The brain on legal highs.
The brain on legal highs.
with Dr. Colin Davidson, St. George's University of London
Hannah - Colin Davidson from St. George's University of London has the role of finding out what legal high substances actually do in the brain. So, how do you do that?
Colin - I work quite close with John. He's just along the corridor and I used to work on just methamphetamine and cocaine, and then met John and he started giving me all these completely new compounds. I test them out on pieces of rat brains, so we want to see whether they have stimulant effects, whether it looks like cocaine, whether it looks like amphetamine.
Hannah - When you say that you're looking at a rat brain, how exactly are you doing that?
Colin - Without going into too many details, we kill a rat as quickly and as humanely as possible and we take what's called brain slices. So they're 0.4 mm thick and I try and incorporate parts of the brain that I'm interested in. In this case, it's usually a part called the nucleus accumbens which is the main brain reward pathway where your listeners have probably heard of dopamine as a neurotransmitter. That's the neurotransmitter most involved in addiction and any rewarding activity like eating or sex. So, I've got my little brain slice that I can keep alive in an artificial cerebrospinal fluid and it get oxygen, it gets glucose. It can stay alive for up to 12 hours or more. And I give a small electrical stimulation to mimic an action potential which is what happens in the brain and the small electrical activities cause release of transmitters. So, I mimic that in this little piece of brain and measure transmitter release and then obviously, I'll take a few measurements, add the drug, whatever drug I'm looking at and see if it increases or sometimes decreases the amount of transmitter I'm getting out and that's a very good indication of whether it's got addictive liability.
Hannah - So, what kind of thing would you see if you added for example illegal drugs like cocaine or amphetamine?
Colin - Well, cocaine would increase the amount of transmitter that comes out by about 4-fold so that the actual amount that comes out is higher and it hangs around for a lot longer. One of the first legal highs I tested was something called Ivory Wave that I got from John Ramsey. I actually found that to have a larger effect than cocaine. I wasn't really expecting that. Cocaine is such a great compound at doing this. Amphetamine and amphetamine-like drugs and we haven't mentioned methadrone yet which is the most famous legal high. It seems to be a bit like amphetamine and ecstasy, but they can cause transmitter release without action potentials which potentially makes them more dangerous ,I think, because it's been shown that amphetamine and methamphetamine, these types of compounds actually cause neurotoxicity in animals. There's a lot of animal studies on that and I did some of those when I worked in America. But also, it's been shown quite recently that human methamphetamine, amphetamine addicts do have problems with their brain such as brain lesions and very long term problems after using them.
Hannah - So, it's causing cell death - these nerve cells - to actually die.
Colin - Especially in the dopamine system and a long time ago it was hypothesized that this cell death might lead to early onset Parkinson's, and recent research has suggested that that is true, that the people who were taking amphetamines or methamphetamine 10 or 20 years ago are getting early onset Parkinson's more often than you may expect.
Hannah - And what about drugs like for example, benzo fury, has I believe been on the scene for quite some time now and it seems to be very popular, and it's legal at the moment?
Colin - Yeah, I was at a conference yesterday in Bath actually, one that John Ramsey was organising, the "Sew Prof," the Sewage Profiling Conference and someone gave a talk there and mentioned that benzo fury is actually one of the most popular drugs at the moment. It has been actually associated with 2 or 3 deaths quite recently. It seems to have both stimulant properties like cocaine or amphetamine, but also has LSD- or trippy-like properties, so you get a double effect from it. My colleague, James Moffatt, has shown us that it's a very potent vasoconstrictor, so it's going to give you hypertension and potentially cause other cardiovascular problems.
Chris - You mentioned that that agent has this hallucinogenic property, but obviously, you can't tell that from a slice in a dish.
Colin - We know that LSD works on the 5HT-2A receptor, so a little bit of protein in the cell membranes which has obviously got a normal physiological function but this is the small protein that LSD and these hallucinogenic drugs actually attach to very strongly and stimulate and cause things to happen inside the cell. So, we've shown and I've got a colleague at Roehampton University, Jolanta Opacka-Juffry, who's shown that benzo fury binds quite strongly to the 5HT-2A receptors. I'd also say that you can also get hallucinogenic effects from these psycho-stimulants and back in the '60s, a lot of people who took amphetamine were misdiagnosed as being schizophrenic. So, if you get massive releases of dopamine, it can cause you to have hallucinations and act like a schizophrenic. So, these drugs like the one I mentioned earlier, desoxypipradrol which is fairly potent has actually caused these sort of effects in abusers, and they've done some crazy things recently.
Chris - What about working out whether or not these drugs are toxic because by measuring the nerve transmitters coming out, that tells you something about what they're doing to the brain, but what about going back to your earlier point about the fact that they will do damage potentially? How can you tell that from your slice experiments? Can you?
Colin - Yeah, I've got a method to measure mitochondrial activities. So, one of their ideas is that some of these drugs might mess up their mitochondria and mitochondria are the small things inside your cells that produce energy, ATP. So, if you run out energy, the cell dies and that's been shown for amphetamine and methamphetamine. It can cause damage to neurons, probably by destructing mitochondria. So, I've been looking at some of these new legal highs using this mitochondrial stain in brain slices, but the ideal thing to do would be to try some long term studies where you may dose an animal with one of these drugs for a few weeks then see what effect it's had. That's the best way to do it.
Hannah - So Colin, once you have this data about how these compounds might affect the brain in terms of causing cell death, addiction and also have psychoactive properties, then what do you do with this data? Who do you tell and then what happens? What's the next step?
Colin - Well typically, we try and publish it in a good journal, but that's quite a long process. So, we always try and disseminate the data to the appropriate people and I have given some talks to the ACMD, Advisory Council on the Misuse of Drugs who would inform the government whether they thought something was dangerous or not. I give talks to various scientific meetings and so on, and get the data out there to the people that need to know.

40:58 - Restricted research on magic mushrooms.
Restricted research on magic mushrooms.
with Prof. David Nutt, Imperial College London
Chris - Researchers might struggle to keep up with new legal high compounds being developed, but scientists face a different problem when they're trying to research illegal compounds because David Nutt, professor of Neuropsychopharmacology at Imperial College London, and he's also President of the British Neuroscience Association, had hoped he could investigate the potential of using psilocybin which is the active psychedelic ingredients in magic mushrooms to treat depression. But he and his team then ran into a mountain of red tape when they tried to set up a trial. Kate Lamble spoke to him about these obstacles.
David - We've discovered a serious obstacle to doing this research clinically and which is that there are two complex sets of regulation that really make life very difficult when you want to do studies in patients. And those are the fact that psilocybin like many interesting drugs are controlled are under the Misuse of Drugs Act, but they're put in a particular Schedule called Schedule 1 which means that they're especially difficult to study because they have especially complex controls. And those controls are arbitrary. They don't relate to the harm of the drugs. So, it's kind of paradoxical. I can study heroin easily because my hospital is allowed to hold heroin, but is not allowed to hold cannabis or psilocybin. This is absurd because heroin is obviously much, much more dangerous. So, the rules are arbitrary. They're old fashioned. They've been stuck in the 1970s and we haven't moved on, so that's the first thing. The second thing is this recent set of legislation called the European Clinical Trials Network which has really screwed up human psychopharmacology in Europe. And that essentially sets a whole series of extra barriers to do in clinical research. Let me give you an example. I can prescribe a drug for blood pressure. Beta blockers have been around for 50 years. If I want to research with it, I have to go through a bureaucratic process which can take up to a year to get all the permissions. They make it virtually impossible to do trials unless you've got a company behind you with hundreds of thousands of pounds because it just costs that much to go through these regulations. So, that's what we're up against, these two great obstacles of the law and the clinical trial regulations.
Kate - You mentioned it's a Schedule 1 drug which I think means, it's got a high potential for abuse and there's no recognised medical use. How does a drug get classified in that way that seems to restrict it from research?
David - Well, it gets incorrectly classified. It's kind of painful to have to explain why this happens, but let me take you through it. In 1960 and 1970, United Nations held worldwide convention to do something about illegal drugs on an international scale. They were about looking at drug harms and they decided that they would create a Schedule for drugs which were thought to be harmful and which had no medicinal use. Our British government went along with it and this Schedule 1 was setup, and then they looked at the drugs, they came across psilocybin and they said, "Oh, well that has no medicinal use. Therefore, it must be Schedule 1." The evidence of harm was very limited, but they just thought that all psychedelics were harmful, so they put them all in Schedule 1. They then put cannabis in Schedule 1 even though cannabis was a medicine. It was a medicine until they put it in SSchedule 1 then it stopped being a medicine. So, there was an arbitrariness to it, a perverseness to it. And then in 2005, mushrooms themselves which were legal in 2005, they were put into Schedule 1 Class A and it was an example we see all the time where politicians will goad each other to be harder on drugs. And of course, everyone thinks being hard on drugs is a good thing. By and large, being hard on drugs is not a good thing for research because it completely stifles research.
Kate - Some people would say that putting it under Schedule 1 and restricting it is because we're worried about protecting patients and these drugs being addictive in some way. Is there any worry with mushrooms or psilocybin that if it's used for depressive patients, it might have an addictive quality?
David - You see, one of the amazing things about psychedelics, they're not addictive. Before LSD was banned in 1964, I think there were 6 trials of LSD to treat alcoholism and there's been a recent meta-analysis of that work and it shows it is as effective as anything else we've ever had. It's anti-addictive. A lot of people go overseas, they go to Vietnam and Thailand to have ibogaine treatment for their addiction. There's a whole body of literature about how the mind-changing effects of psychedelics can be useful for addiction as they may well be useful for depression and for dysthymia and all the other things that people get benefits from them with.
Kate - We're now seeing a lot more research into how class A drugs, as we see them quite illegal drugs like ecstasy being used for PTSD and things like that, and these drugs having medical implications. Do you think that that legislation is holding us back, or do you think that there is a certain amount of caution that we should be using ecstasy?
David - I mean, most of the drugs in schedule 1 are nothing like as harmful as the drugs in schedule 2. It's an arbitrary distinction. They're there not because of their harms. They're there simply because someone said that they don't have medicinal use. And it becomes a circular argument. If it's impossible to study them as it is almost impossible, only very cussed people like me can be bothered to pursue the years and years of hassle trying to get licenses. There's no question that if the scheduling was more rational, there'd be a lot more research and that will be good for neuroscience and that will be good for patients. The worst example is not psilocybin. It's cannabis. I mean, cannabis was a medicine. It should never have been put in Schedule 1 and exploring that has been terribly slow over the last 40 years because of it. And it probably means that lots of people have suffered unnecessarily because of the use for spasticity, the use for pain. And also, there's a huge interest in cannabis-type drugs for cancer treatments and all these has been really impeded by the scheduling. And of course, it hasn't actually stopped young people using it. When cannabis was put into schedule 1 in 1971, half a million people in this country had ever used cannabis. Forty years on, 10 million people have used it, probably more, probably more like 20 million now. So, it hasn't stopped the use. All it's done is stop scientists using it.
Kate - As we're discovering more medical consequences from these drugs which we've previously been restricted of using, do you see a point at which scientists will campaign or win a change in regulation for research purposes?
David - Well, I am pursuing a campaign now to do that. I want scientists to sign up. I've written over to a couple of colleagues quite a powerful piece coming out in Nature Reviews Neuroscience in June that argues the folly of what we've done for the last 50 years and that strongly challenges the government to change. So, I want scientists behind me because I think when they see the missed opportunities, they'll realise that neuroscience hasn't expanded, particularly in fields like consciousness and pain research. There are enormous insights to be made from drugs like psilocybin and cannabis.
Kate - Let's say that you finally get the trials under way. What's your foresight of when we could find out whether this can be turned into an effective treatment?
David - The current plan is this, get the trials started as soon as possible. If there is significant benefit, then obviously, we want to find a way of making it available to psychiatrists. And this is why the law must change because I can tell you that under the current law, psychiatrists would need to have a license to use the drug. The license costs 6,000 pounds and takes a year to get. So, they're not going to use it. Hospitals need to have a license to hold the drug. There's only 4 hospitals in the country with that license. So, we must change the law to anticipate benefits of these drugs so that patients can then have access to them.

49:15 - Is sleepiness related to cheerfulness?
Is sleepiness related to cheerfulness?
Hannah - Is there a relationship between being an early riser and being chipper? We turn to the sleep clock expert, Dr. Mick Hastings from the Medical Research Council in Cambridge. He had some follow up questions for Stuart and it turns out that grumpy family members are still grumpy even if they get to have lines and adjust their sleeping patterns during long holidays. So, it doesn't seem to be the case that the bad mood was simply due to going against their midnight owl body clock cycles. So, given it's not natural body clock differences, what could be going on in the brains of these different groups? Michael - Well, this is where it gets more biochemical, but I'd argue even more interesting. We do know that how well we feel, our mood, of course, is determined by the chemicals in our brain, the neurotransmitters which are active. And we also know that the body clock controls the activity levels of those neurotransmitters. There's one in particularly called serotonin which is also called 5HT which is a sort of happy neurotransmitter and quite a number of drugs which are used to control people's moods act through serotonin and the receptors it works upon in the brain. So, one possibility could be, there's just something different in the way in which the body clock controls serotonin patterns in the grumpy and happy people within this family. Something else we could look at where we know the body clock has a direct effect on mood is through a hormone called cortisol. Now cortisol is very important because it energizes the body, sends sugars into the bloodstream, increases heart rate, muscle tone, and what have you. So, it's the hormone that's secreted under the control of the body clock, just before we wake up. Which is great, you jump out of bed, you're ready to go. Perhaps there's something different in the cortisol patterns. We know they do vary enormously between people because an important feature of cortisol is that not only does it energize the body. It does have a suppressive effect on mood. Now, of course I'm not a clinician, I'm a basic biologist, but an interesting experiment would be to take some saliva samples from individuals, measure their cortisol levels and just see if there's a correlation or relationship between how grumpy people are in the morning and their morning levels of cortisol. Hannah - So, possibly changes in the serotonin or cortisol levels in the brain could be affecting these morning moods. In this case, is there anything that Stuart's wife and grumpy son can do to cheer themselves up in the morning? Michael - I think it's important to think about what might make them happy. Perhaps they could give themselves some little treats to wake up to. You know, a hot cup of drinking chocolate or the sunlight shining on the bedroom wall.
- Previous Measuring the Universe
- Next Happy Birthday DNA - Genetics Society










Comments
Add a comment