eLife Episode 56: Vaccines and viral swarms
How the brain handles sensations from amputated body parts, evidence that government vaccination campaigns to target measles really work, the heel-prick blood test at birth that can detect prematurity, testing the reproducibility of science at the level of a whole nation, and the multipartite viruses the spread as an infectious swarm: scientists show that they replicate different parts of the virus in different cells...
In this episode

00:38 - Amputation, brain changes and phantom pain
Amputation, brain changes and phantom pain
Daan Wesselink, University of Oxford & UCL
Different regions of the central nervous system are specialised for performing certain tasks. And in areas like the somatosensory system, for example, the nerve cells are organised into a map that represents the body surface. And a stimulus delivered to any part of the body produces activity in the corresponding brain area. So what happens if someone undergoes an amputation? Does the brain still incorporate the now-missing anatomy in its neural map? And how does that compare with people born without that same body part in the first place? Chris Smith got the answers from Oxford University's Daan Wesselink...
Daan - A few years ago we had invited an amputee who had very vivid phantom sensations. And what I mean with phantom sensations is that, even though they'd lost their hand a long time ago, they could still feel their phantom hand as vividly as their other hand that they still had. And when we asked him in a scanner to move their fingers - that they don't have - their brain was activated exactly the same as this other hand. And we then wanted to do a big study where we compared every type of one-hander, both feeling their phantom out very vividly, and not feeling their phantom at all.
Chris - Now we've been brought up that practice makes perfect, and if you're a fine musician, or you learn another language, that actually this leads to remodelling of the brain - the cortex - so that we can generate more connections and have that ability. Having a phantom symptom that won't disappear kind of flies in the face of that, because it says the opposite is true; that once your brain has gotten an established map of a part of your body, it doesn't get rid of it.
Daan - Right. And I think both things are true: so the brain is able to save some information it's learned at a very young age and keep that going through the rest of the life; but also is plastic, and able to adapt through the things we learn. So with this study we really looked at the most basic area in the brain, dealing with touch, and say that that area...actually the fine details might be slightly different, but the overall architecture just stays the same once you've developed a hand.
Chris - How did you reach that conclusion? What was actually involved in doing the study?
Daan - We’re a neuroscience lab, and what we do is we use fMRI, which is basically this large machine in the hospital where we put people in. And then when they do a certain action in this scanner, we can see how active these different brain areas are. And so we were particularly focused in the touch area. So we put two groups of participants inside the scanner: so both people who had lost their hands later in life, and people who are born without a hand. And then when we looked at this brain area, which in you and I is a hand area, there was nothing there in people who had never had that han. And that's not so surprising, because they never had that hand. But then when we actually looked at the people who also didn't have that hand, but did have it when they were children, we couldn't distinguish it from that of people who still have both hands.
Chris - What that's telling you then is, as you surmised, that when you have a hand to start with, the brain establishes a map and a representation of that body part. If you then lose that body part, the brain doesn't magically remove that representation. It stays there, and is still capable of creating a mental image for that person of the part of the body that no longer have. So this must be the underpinning of when people have say, phantom sensations, it's because that brain representation is still there.
Daan - Yeah, exactly. We think that this somehow is produced by this area that still has all this information. However, we were even more surprised when we also put amputees, people who have lost their hand later in life. But some amputees do not feel this phantom hand anymore, they just lose it as well. But even in these amputees, we could find all the hand information in this brain area.
Chris - Now what do you think contributes to the maintenance of that representation? Is it just that, once you've laid down a patch of brain architecture when you're young, it's locked in and you're stuck with it? Or do you think that, because the sensory system has these reciprocal connections to many other regions of the brain - both the motor areas, so that you can anticipate what you're going to feel when you move, for example; also, the equivalent motor area on the opposite side of the brain, because the two reciprocally linked, aren't they. Do you think that it's the inputs from those other brain regions which helps to maintain this map?
Daan - Yeah, it must be somehow that the brain is able to keep this input from other areas. We're looking into it right now with more experiments. The only thing we know: it's not the other hand. So the two hands are in that aspect sort of independent. But like you said, motor areas could be true.
Chris - So why do you think, then, that some people do have more vivid phantom sensations? Why is it that people who don't have that appear to be better at remodelling their brain?
Daan - There are some behavioural experiments suggesting that what is important is not necessarily this most fundamental brain area, dealing with touch, creating sort of a map of the body, but some other type of readout. That there is another brain area that reads whatever information is in that area, and that brain area just stops reading it. and people lose this phantom sensations. But it's hard to say for sure unfortunately at this point.
Chris - Essentially what you've managed to do here is to confirm what we suspected might be going on, which is you've got this fairly fixed map, which is established early. Because we had clues that that was there from the days of the visual system, didn't we, with the Nobel Prize winners Hubel and Wiesel who actually did similar experiments in animals, showing that if you deprive the animal of visual input from a very early stage, you get permanent changes to the architecture of the nervous system. So that sort of agrees what you're showing here. But what can you now do next in order to take this further?
Daan - Right. I think the motivation really to move forward now is in neuroprosthetics. There's been a lot of research going on, on how to get this information from the brain to some kind of mechanical device that will replace a hand, and there's sort of two teams here. One group of engineers, they try to just access the brain, and try to have the controls make somehow sense to you and I - just be as natural as moving a real hand. And other people try to use other types of information, maybe like movement of the arm muscles, or movement of the eyebrows, that then will control this robotic hand. And I think we really like to say, well, in these people who have lost their hand later in life, all this information is still there, so you should probably use this information in the brain. Whereas in people who have never had this other hand, you would need to use other types of information. So other types of controls...

07:21 - Do measles vaccine campaigns work?
Do measles vaccine campaigns work?
Prabhat Jha, University of Toronto
Before the 1980s when mass vaccinations began, measles killed nearly 3 million people - most of them children - every year. Where vaccines are rigorously used, the numbers of deaths are now very low. But, in some places, uptake has been more patchy and also the absence of actual data proving that vaccination campaigns save lives have made them harder to implement, particularly in resource-poor settings like India. Speaking with Chris Smith, Prabhat Jha, from the University of Toronto, has used data from a mortality survey in India, called the million death study, which he leads, to compare measles fatality rates in areas of the country with vaccination campaigns and those without…
Prabhat: Do measles campaigns save lives? This was the central question we sought to answer in India where, measles deaths remain quite common. And what we found is that the government of India's national measles campaign avoided something like 50,000 deaths over a three year period. So this study very clearly answers that question, that special efforts to eliminate measles deaths work where measles deaths remain common.
Chris - I must admit Prabhat, when I read your study I was pretty surprised to learn that we don't actually have data supporting doing these sorts of campaigns. It seems a little bit like it's assumed knowledge.
Prabhat - You're right, Chris, that public health has acted on measles without information on whether it actually made a difference. But we know from all sorts of experience, including our kids being immunized against measles in high income countries, that it does save lives. Just the recent controversy and cases that have been reported in the US, where measles immunization has tailed off, is a demonstration of that. But the big picture globally is of deaths from measles in places where it remains common, such as in India or parts of Africa. And importantly, in these settings, most deaths don't occur in the hospital; but rather they occur at home without medical attention. So the million deaths study obtains a random sample of all of the deaths. And in this case we studied in particular 27,000 child deaths from 2005 to 2013.
Chris - And what's the significance of that date window?
Prabhat - The significance is that in around 2009, the government of India launched a special campaign to try to reduce measles deaths, in the states of India where measles was more common. And what we were able to do is to see a reduction in measles deaths in the places where the campaign had started.
Chris - In essence then, if you're looking at a range of different causes of mortality, and you see a reduction in measles, but other things like diarrhea for example remain stable, you can be reasonably sure that the intervention - i.e. the vaccination campaign - has had something to do with that difference.
Prabhat - That's absolutely right Chris. What we did is look at whether the decline was greater in the special states, that campaigns states, and it was - it fell by about 27% measles deaths in those states, versus the non campaign states where it fell 11%.
Chris - So it obviously is achieving what it sets out to do. If you look in more detail, though, at who it's saving and who it's helping, is it across the board? Or are there any exceptions or unusual outcomes from it?
Prabhat - What was welcome news in this study is that the reduction in girls’ mortality from measles was greater than boys’. Now generally, mortality from measles in India is higher in girls than boys. And we also found that particularly in the districts where the campaign was most active was the biggest reduction. So it does suggest that the campaign - which doesn't rely on families bringing their children to a clinic, but rather goes out to the house, finds unvaccinated children or under-vaccinated children, and whacks them with a second dose of measles - was actually quite effective.
Chris - Why do you think this study is actually necessary though? Because could the WHO have not told you that, just look at the data for any country, and if you vaccinate against this disease you get lower mortality rates. It saves lives, this vaccine, therefore it's a good idea; and it doesn't matter which country you live in, you're going to benefit.
Prabhat - There's two reasons why we need direct evidence. The first is that the WHO or model-based estimates are probably overestimating the numbers of actual measles deaths. And the second is local believability. Governments and media, and particularly the public, can be skeptical of sometimes foreign labelled models. They think, “oh well, these are just do-gooders from outside.” And in India, like in the UK or in Canada or the US, there is a strong anti-vax movement. So getting good local data that are generated by the government, rigorously evaluated, and saying, “well, these are your own numbers that are showing that measles vaccine saved lives,” is important for the public discourse. It will change minds, we hope.
Chris - Have you sent your results to Mr. Modi, and said, “well done, have a pat on the back for a campaign well done, now make sure you keep funding it?”
Prabhat - My approach is to always rub the nose of politicians in the data and nothing else. We had co-authors from the Indian government, and they've expressed a strong interest in saying, yes, they would like to think about potentially a measles death elimination effort. The hope is that they will also implement more decentralized simple systems like this to count the dead and describe the causes, and then they can relate it to the amount of spending that is ongoing, and use that mortality surveillance as a way of monitoring the progress. It's good news, and we hope there will be more.

14:19 - Heel-prick test for prematurity
Heel-prick test for prematurity
Kumanan Wilson, Ottawa Hospital
But first, to childbirth. The average human baby takes 40 weeks to develop from the time of conception to the time of birth. And in developed countries, a dating ultrasound scan, included as part of the antenatal screening process, establishes when the baby’s due. So if an infant arrives early, doctors can tell and look out for appropriate problems accordingly. But in poorer countries, this sort of information may not be available. And under these circumstances, if a baby’s smaller than it should be, is that because it’s premature, or is something else wrong? Previously we had no idea. Now, speaking with Chris Smith, Kumanan Wilson, from Ottawa Hospital, describes his possible solution…
Kumanan - We've developed a blood test that can be done on an infant soon after birth, that can tell us whether that child is premature or not. Premature births are one of the leading causes of death and disability in children around the world, and this is particularly a problem in low resource settings. Expectant mothers don't have access to the type of prenatal care we have in more developed settings, and they don't have access to prenatal ultrasound in particular. So when a child is born, we don't know whether that child is born too soon, or premature, or born on time.
Chris - So how did you actually do the study?
Kumanan - This study is a validation or a further test of something we developed on children born in Ontario. Our first study developed a mathematical model that could look at blood from a prick of the heel of a newborn infant, and could tell you how premature that baby is.
Chris - So when you take the heel prick sample, what do you look for in the blood in order to make that determination of whether that baby was born on time or not?
Kumanan - This blood sample we obtained from the child is what we do in a routine newborn screening program. And in that program we test for a variety of illnesses, such as cystic fibrosis, or thyroid disease, or things like sickle cell disease. For the purposes of the newborn screening program we look at things like amino acids, fatty acids, haemoglobin levels. We've known from past studies that these vary based on the gestational age of a child. What we were interested in finding out was, if we had the patterns of those chemicals, could we do the reverse, could we determine the gestational age of a child? And that's why we were able to successfully do in our Ontario sample of children.
Chris - This is what researchers dub "metabolomics", isn't it? Where we take a combination of markers, and we say, "when you see these markers present at these levels, this can be used to make various predictions".
Kumanan - That's correct. This is a form of metabolomics. In this case we're using a convenient sample of chemicals that we obtain from our newborn screening program. It is possible we can make this test better if we expand and look for chemicals that we aren't routinely collecting.
Chris - Now when you subject the samples to your analysis, how accurate is it? In other words, if you take your gold standard infants that have been ultrasound screened, do you know what their gestational age is when you take the sample? How out is your prediction versus reality?
Kumanan - We are able to predict the gestational age within one week. However the reality is, in the vast majority of our sample, the children were born at term, and birth weight alone would have been adequate to determine the gestational age of those infants. Where the added value of the metabolomics is in children who are small. In children who are born small, there is the risk of misclassifying them as premature, when they may be small for other reasons. For example, they may be sick.
Chris - So this is against a gold standard population of individuals born in a rich country, first world setting. Have you validated it against individuals who were born in the target territory? So third world settings, or resource poor settings. And also, might the levels have a degree less of confidence when you take into account ethnicity?
Kumanan - These are great questions. The first study we did after we developed our test in Ontario was to use our immigrant database to look at different ethnic groups. And we found that within our Ontario cohort, the test performed well amongst the various ethnic groups. The study that was published in the eLife journal then further validated this in a cohort in Bangladesh. This is a cohort that is part of an ongoing study. We were fortunate because they had dating ultrasounds done on all of the infants. And we found that the test we developed in Ontario performed very well in the infants born in Bangladesh. That increases our confidence that this approach can be used in multiple settings, in multiple low-resource settings, and can help us determine both the burden of preterm births, as well as help us evaluate interventions that are designed to reduce preterm birth.
Chris - And is that where you're going next with this then?
Kumanan - The next step is a partnership we have embarked upon with Stanford University to expand this approach to multiple lower-resource settings. In Zambia, Zimbabwe, Kenya, and another site in Bangladesh.
Chris - And to throw you a bit of a curve ball, are there any situations, or are there any diseases, acquired conditions, inborn conditions as well, which can throw your system? So in other words you get data that look promising for the individual, but actually you're being misled?
Kumanan - This is actually one of the questions we want to answer as we move forward. We do wonder if a stressed sick child at birth, that can distort their pattern of chemicals, and that might affect our assessment of gestational age. So that's a very important question for us to answer. We hope to explore that as we examine the samples from these multiple sites.

20:53 - Science reproducibility in Brazil
Science reproducibility in Brazil
Olavo Amaral, University of Rio de Janeiro
In recent years a number of initiatives have been launched to test the reproducibility of published scientific data. Mostly they’re operating within specific scientific domains, like the cancer reproducibility project we reported on back in 2014. Now, speaking with Chris Smith, Brazilian researcher Olavo Amaral is going a step further and launching a reproducibility initiative that, for the first time, aims to assess performance at the level of his whole country…
Olavo - We're trying to get as much of a representative sample as we can of Brazilian biomedical science. We're from Brazil, and we're interested in how the country is doing, so we're taking a random sample of Brazilian articles from the last 20 years in the life sciences; constrained by method, because of course we have to do things that our lab network can perform, to actually repeat them in multiple labs to see how reproducible the original finding is.
Chris - How many laboratories are going to repeat each of the studies?
Olavo - We've put out an open call for laboratories within Brazil. We currently have more than 70 labs actually engaged in the initiative. The idea would be to have each results replicated in at least three laboratories, so we'll get a feeling of the intra-lab variation.
Chris - So how is this going to lead ultimately to an improvement in standards of science, both in Brazil and also internationally? In other words, what are you going to do with these results once you've got them?
Olavo - Yeah. Well we hope it will. We can promise. But I think I think first of all it's important to talk about the subject. Just actually having the project happening, I think, does make a difference in raising the debate about the problem. We've been having reasonable media exposure in Brazil, so just having a lot of people discussing this I think is already a plus. Once we have the results: one, we can get a better estimate, what is our responsibility? It is hard to compare, because it's not like we have a lot of statistics from other countries, or be like the first country to actually try to do that nationally. But I think we can also try to assess what predicts responsibility, what kind of findings are more reproducible, what findings are less. Are there features of the study of the method of the researcher that count, and can help predict what is reproducible, what is not? And I think that is actually very useful for agencies that fund science, for institutions who develop science, to try to actually have a better way to assess the literature that does not count only impact, how much a paper’s cited, but also what are the chances that this finding should be reproduced in other laboratories.
Chris - Because obviously funding is a sensitive subject in all countries, but it's especially a sensitive subject in Brazil, because recently, hasn't the government passed some kind of legislation suggesting that you're going to be on the same scientific spend annually for the next 20 years?
Olavo - It's only getting worse. We have a cap on public spending for the next 20 years, but we're actually getting cut. The funding for the Ministry of Science and Technology has just been cut around 40% a few weeks ago. Funding is about a third of what it was some years ago. So I mean, it's really unpredictable in terms of how it's going, and to us it is a very sensitive issue. On the other hand, I think it raises the question that we have to spend as wisely as possible. So I think it's important to study ways to better direct this funding.
Chris - And when's the study going to report? When are you aiming to be finished by?
Olavo - Probably in 2021. We're starting experiments in the second half of the year. There is a lot of labs, so they'll have different rhythms. Each lab is supposed to do between three and six experiments. I suppose that will happen over the course of a year, maybe, and it will probably be late, perhaps early 2021.
Chris - And given the parlous political and funding situation in Brazil at the moment, is this a securely funded initiative? As in, you're not going to get halfway through this, and then half the labs get their funding pulled, and the project never gets finished?.
Olavo - We actually have private funding for this. We're funded by the Serrapilheira Institute, which is a private funder which aims to fund science in Brazil. We do have the money already. We are not exactly sure how many experiments we can do with it, because I mean we still have to figure out some logistics and some costs, but we were pretty sure that we can do at least 60 experiments in three different labs Our aim is to get to 100. We'll have to see if our budget goes that far.
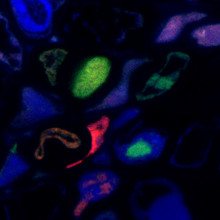
25:21 - Viruses that spread in swarms
Viruses that spread in swarms
Stephane Blanc, University of Montpellier
The majority of viruses that we encounter consist of a single particle that contains all of the genetic information required to stage an infection successfully. But that’s not true of all viruses, especially some of those that crop up in plants, and Chris Smith hears about one such "multipartite virus" - faba bean necrotic stunt virus - that replicates different bits of the virus in different cells from the University of Montpellier's Stephane Blanc...
Stephane - These viruses are called tripartite, which means that each piece of information is packaged separately in a distinct virus particle. So what you call the virus is in fact a small population of virus particles. The whole information is never contained in a single virus particle for these viruses.
Chris - That’s Stephane Blanc and, yes, you heard that correctly - the virus he studies - called “Faba bean necrotic stunt virus” - effectively operates as an infectious “swarm” where different viral particles contain different genetic elements and they have to share the resources that each encodes and produces to enable all of the viral particles to replicate and sustain the population as a whole. Obviously this comes at a cost…
Stephane - Theoretical studies concluded that if the virus has more than three or four pieces for the genome information - and the virus we work with has actually eight - and they concluded that these viruses should not evolve. This should not work. So that was the basis of our whole program, that we work with a virus that should not exist.
Chris - Indeed, if you’re relying on gene products from another viral component that might not even be in the same cell as you, that doesn’t sound like a recipe for success, does it.
Stephane - This conclusion that it should not exist is based on a strong assumption: that all the pieces must be together within a cell for the system to work. So we decided to just verify this. Are all the pieces together in an individual cell. Or is this just a sort of dogma that may not be true in our case? We decided to localize each individual piece in an infected host. And for this, because each piece of the genome has a different sequence, we can make probes that are specific for one piece. Those probes can be fluorescent with different colors. And then we can see whether the colors are always together, or whether the colors are separate; which would mean that the different pieces do not need to be together.
Chris - But this virus is split up into 8 distinct, independently-replicating pieces, and with present technology it’s not easy to look for all 8 at the same time. So Stephane’s solution has been to look for pairs of genome components that are present in the same cell to work out how likely it would be statistically to end up with all 8 pieces in one cell at the once.
Stephane - If one piece of genome is together with another piece of genome in, let's say, 30% of the cases; and that this other piece of genome you can show in another experiment that is present with, again, another piece of genome in 30% of the cases; then you can calculate the probability to find a cell with all 8 segments. So this is a statistical inference. And this type of estimation predicts that we probably have 1% of the infected cell that may contain all eight segments. So they are extremely rare.
Chris - In fact, they’re far too rare to sustain the population. And it’s not just a copy-number effect where the team are seeing cells with a detectable amount of just a couple of genome segments at one point in time: they’ve followed the infection for hours in these cells and never seen other genome segment signatures develop later.
Stephane - All the cells contain some piece of the genome. And these pieces of information, although they are not with all the pieces, they are replicated. So it means that in those cells where the genome is not complete, the genetic information is replicated. So the replication cycle occurs even in cells where the genome is not complete. And that's the discovery.
Chris - So a cell infected with just part of the virus can nonetheless still replicate that component. But this would require components made by bits of the virus that the cell doesn’t actually have! So how does it do it?
Stephane - What we call the piece of information of the genome. They are genes that produce something that the virus needs to build baby viruses. So in different cells, different pieces of information produce this material. and it must be exchanged in between the cell. So that's what we believe, that the virus, one piece of information is sending away its product to complement the other pieces of information in other cells. And for this, either the virus is opening gates, communication between cells so that it can exchange product between cells containing different pieces of information; or the virus uses natural cell-to-cell communication of the host. This we don't know yet. We have to investigate the details of the mechanism in the future.
Chris - So there's still work to do to establish how the population of infected cells are networked together to share resources in this way. There's also the obvious question of why does this virus do this at all.
Stephane - Many viruses are in just one piece. So these are the canonical view we have on viruses, derived from the canonical viruses, which are in one piece. No-one clearly understands why these viruses in several pieces actually do it. But our contribution is that before our contribution, people in the community were wondering, how is this possible, right. Because they cannot efficiently bring all pieces together in an individual cell. We show that they don't have to do it and that the system can function. So what we address is more that the cost of doing that is not so high. But we do not address that benefit. So the benefit remains a mystery.
Related Content
- Previous How to see faint stars
- Next What causes cyclones?










Comments
Add a comment