The body clock goes under the Naked Scientists' spotlight this week. We unpick the mechanisms that enable human cells, plants and even bacteria to track the time of day and alter their activities accordingly, and we hear the evidence that night work makes you put on weight and boosts your diabetes risk. In the news, how cells grafted into the eye restore sight to blind mice, the three genes that can convert scar tissue back into beating cardiac muscle following a heart attack, and electrical stimulation that returns movement to limbs paralysed by spinal injury. And on the subject of the body clock, can an e-book at bedtime keep you awake at night?
In this episode
- Nanoparticle drugs for Cerebral Palsy
Nanoparticle drugs for Cerebral Palsy
Drugs attached to nanoparticles reduce the symptoms of Cerebral Palsy in rabbits if given soon after birth, by getting into the brain and targeting just the cells involved in the disease. This opens the door to treatments for a range of neuroinflammatory diseases including Alzheimer's disease and multiple sclerosis.
Cerebral palsy is an umbrella term for a suite of disorders, all of which stem from an injury or insult to the developing brain and affect muscle coordination, cognition and sensory acuity. Once Cerebral Palsy is diagnosed, it's usually too late to treat the brain injury itself, so current treatments concentrate on easing the symptoms.
It's thought that two cell types are involved in the pathology of cerebral palsy - Microglia and astrocytes. Microglial cells are part of the immune system that police the brain and spinal cord, and in Cerebral palsy are thought to be the cause of inflammation. Astrocytes are essentially neuron support cells, but fail to protect nerve cells in situations of inflammation.
Writing in the journal Science Translational Medicine, Sujatha Kannan from Wayne State University and colleagues describe how these cells, normally protected by the blood-brain barrier, can be targeted by attaching drugs to branching nanomaterials known as dendrimers - which are already being investigated as a targeted delivery mechanism in cancer and other diseases. The drug they were using was NAC (N-acetyl-L-cysteine), a known antioxidant and anti-inflammatory agent which is also being tested as a treatment for Alzheimer's and Autistic spectrum disorders.
The drug, linked to its dendrimers, was delivered to new-born rabbits that showed neuroinflammation and motor deficits, a good model of human cerebral palsy. A single dose led to a significant improvement in brain injury and motor function, whereas the drug without its nano-partner did not, suggesting that the dendrimers are key to getting the drug to where it's needed.
This shows that there is a potential window for treating the brain injuries that lead to cerebral palsy, but we therefore need better ways of identifying those at risk early on. According to the authors, "targeted therapy ... in at-risk patients, delivered at an early stage after birth, can potentially arrest or prevent the development of motor and cognitive deficits associated with perinatal brain injury and cerebral palsy."
This is also a good test of nanoscale dendrimers as drug delivery agents, and gives us good reason to look forward to the results of other trails testing these in a wide range of diseases.

01:36 - Setting the circadian clock
Setting the circadian clock
with Professor Russell Foster, Oxford University
Chris - The ancient Greeks are credited with first pointing out that some plants appear to be able to tell the time by altering their leaf shapes during the day and some of the first evidence of the existence of a similar internal clock in animals, including in us, was produced about a hundred years ago when scientists showed 24-hour patterns of activity even in the absence of any time cues. Now we know a lot more about how this whole system works and that's partly thanks to one of the pioneers in this area of study which is Oxford University's Professor Russell Foster who's with us this week. Hello, Russell...
Russell - Hello, Chris.
Chris - Thank you for joining us. First of all, how does this body clock actually keep time?
Russell - Yes. First of all, each of those individual SCN cells are talking to themselves and that coupling is really important in part of the 24-oscillation and then there are two ways in which the body clock will communicate with the rest of the organism. One is via direct neural connections and there are other humoral routes; some sort of neurohormone release from the SCN. Really, that's where our understanding is not so good because we don't really understand in fine detail how the master clock communicates with the rest of the body.
Chris - It's one thing for a bunch of nerve cells to have an activity like this. It's another for other cells around the body to pick up on and respond to that if they don't have a direct nerve connection.
Russell - Yes and what we used to think is that this master pacemaker simply imposed and drove a 24-hour oscillation on the rest of the body. Then, from Schibler's group in Switzerland, he showed that individual cells could still regulate - not SCN cells - in fact, these were fibroblasts. They've been in culture for 30 years.
Chris - Skin cells.
Russell - Yeah. He shocked them by putting 50% serum in the medium and then showed that there was gene expression with a pattern of 24 hours, showing for the first time that essentially, every cell has this capacity to generate this 24-hour rhythm. What's so exciting is that the SCN has to act as this master pacemaker, rather like the conductor of an orchestra producing a rhythmic signal from which the rest of the body takes its cue.
Chris - What is the nature of the actual clock work? What are the cogs in those nerve cells that are ticking around, keeping time?
Russell - The molecular clock. It's fundamentally a feedback loop whereby a gene produces a protein, interacts with other proteins to form a complex and then enters the nucleus, and then regulates its own expression. So you have essentially a molecular oscillation.
Chris - A sort of genetic domino effect, one thing turns on, the next trips on the next, turns off the first, and it goes around in a circle that happens to take 24 hours.
Russell - Very much so. And of course, the rate at which you turn those genes off, the rate at which the proteins are produced, the rate at which they form a complex, the rate at which they enter the nucleus, all of those things interact to form the 24-hour dynamics. Therefore, you need a whole range of other proteins, sort of kinases, regulating the activity of the protein complexes.
Chris - What about the fact that we don't live in a world where time is static in the sense that I can jump on an aeroplane, I can go forward and backwards in time zones, because of the latitude we live at, we have days which are longer and shorter at certain times of the year? How does the clock accommodate that?
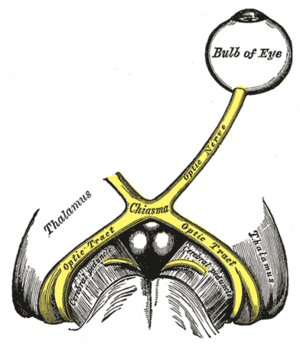
Russell - Well, the clock, to be of any use in actually fine-tuning physiology and behaviour to the varying demands of the rest-activity, light-dark cycle, has to be set to local time. The critical mismatch when one travels, let's say, from Cambridge to New York, is that you need to realign to New York time. The primary, but not exclusive way, that the body does that is exposure to the light-dark cycle. The new light-dark cycle sets the internal master clock in the SCN to local time, and we know that it's the eye that's detecting that light in us and there's a special cell within the eye that's detecting this light. So for example, it's not the classical visual system, the rods and cones of the eye, that are required for this light detecting mechanism, but another cell class.
Chris - Go on. I'm intrigued. What is the cell?
Russell - Well, it's a small group of photosensitive ganglion cells. Now the ganglion cells are those cells in the retina that are almost the last bit of the pathway. They're the cells that form the optic nerve and then go off into the brain. What's become very clear over the past 15 years, I suppose, is that about 1 in 100 of those ganglion cells is directly light sensitive and will project to the clock structures in the brain, the SCN.
Chris - So, you are exposed to bright light, these ganglion cells interpret the bright light, and they tell the brain about it, so the brain has that time of bright light exposure. It knows when morning is and it uses that to refine the clock.
Russell - Absolutely. What's turned out to be completely remarkable is that you can be visually blind, have no conscious perception of light, and yet these photosensitive ganglion cells are still there, working away, regulating your internal time. We've studied several individuals now who have lost their classical visual cells, they've lost their rods and cones, and yet, they can regulate their body clocks perfectly well.
Chris - What happens if you have individuals who have no eyes for one reason or another, or they've lost their eyes through injury? Would their body clock fail to reset in this way then?
Russell - Absolutely and this is why we're working with our colleagues in ophthalmology, because ophthalmologists not only need to explain what it's like to lose your vision, but in those circumstances where you've lost your eye, they also need to know what it's like to be plunged into unremitting jet lag for the rest of your life.
Chris - Sounds awful.
Russell - It's terrible. And of course, jet lag is so ghastly, not because you're simply 5 hours let's say, shifted from Cambridge to New York. It's because we were talking about the mismatch between the internal clock, the master clock in the SCN, and all those peripheral clocks. It's as if you've got a temporal smear. Everything is slightly misaligned. It's rather like the conductor is at a different beat from the violin section to the brass, the whole cacophony of sound rather than a symphony.
Chris - It really does sound awful. So all of your tissues are effectively running on different time zones and so, there's a communication problem between them, which is why you feel so awful when your jet lagged but that could be at the root of diseases where the body clock doesn't work properly there.
Russell - Absolutely and that's the thinking. That in fact, it's internal desynchronisation which is such a problem for overall health and it may be one of the problems that for example, night shift workers get.
Chris - What about people who have other disorders, mood disorders, psychiatric disorders, bipolar disorder where people can't go to sleep, depression where people struggle to get to sleep, and then they wake up very early? Is the sleep disturbance a cause of their symptoms or actually, is it just one manifestation of the underlying process?
Russell - In severe mental health, in psychosis for example, it's been reported that abnormal sleep has been recorded way back from the 1880s, but it's always been assumed more recently to be, well, you know, individuals with mental health problems don't hold down a job, or the antipsychotics they're on, and the bad sleep is a consequence of that. What we're discovering is that that's not right. There seems to be a fundamental overlap between the mechanisms that predispose you to poor mental health, and those that are generating a normal sleep-wake cycle. What's become very clear is that of course, sleep is a lot more complicated than just the body clock. It's involving multiple brain structures and multiple neurotransmitters. A defect in one of those, that gives rise to a mental health problem, will also have a ripple effect across sleep and sleep problems.
Chris - Brilliant! We'll leave it there for now. That's Professor Russell Foster, he is from the University of Oxford.
And if you'd like to see how the body clock works, we've put together a special Naked Scientists scrapbook episode for you this week, which takes you through graphically how the circadian clock works.

10:35 - Body Clocks, Obesity and Diabetes
Body Clocks, Obesity and Diabetes
with Dr. Orfeu Buxton, Harvard Medical School
Ben - Obesity is a growing problem in the western world, to the extent that it's being dubbed an epidemic. The concern is that it's linked to diabetes, heart disease, stroke, and a wide range of other conditions. But why are obesity rates rising so rapidly? New research suggests that poor sleep may be partly to blame, as Hannah Critchlow has been finding out.
 Hannah - Our worlds are becoming increasingly 24/7. About 20% of the working population - so about 1 in every 5 people - work non-standard time, so not your usual 9:00 to 5:00. People are also regularly travelling across multiple time zones for work or pleasure. And on top of this, about 30% of adults report one or more of the symptoms of insomnia, so difficulty getting to sleep or difficulty staying asleep.
Hannah - Our worlds are becoming increasingly 24/7. About 20% of the working population - so about 1 in every 5 people - work non-standard time, so not your usual 9:00 to 5:00. People are also regularly travelling across multiple time zones for work or pleasure. And on top of this, about 30% of adults report one or more of the symptoms of insomnia, so difficulty getting to sleep or difficulty staying asleep.
At the same time, obesity rates are on the up. Here in the UK, over a quarter of adults are obese and 3 in 10 boys and girls aged 2 to 15 are either overweight or obese.
Diabetes is also on the rise. In the UK, the number of people diagnosed with diabetes has increased from 1.4 million to 2.6 million since 1996 to 2010. So, are these increased problems linked?
A new study by Dr. Orfeu Buxton and his team at Harvard Medical School has uncovered the physiological link between lack of sleep, changing circadian rhythm, diabetes, and weight gain.
Orfeu - What we did was schedule the participants to a 28-hour day. So, each day, their light and dark cycle, their sleep and wake, and their meal timing was shifted to 4 hours later. This would be similar to being on a planet with a 28-hour day. Our internal circadian clock can't synchronise to such a schedule. Imagine the last time you had a large meal at 2:00 in the morning. Your body is just not ready to process that food and our participants experienced that for 3 weeks. That was the circadian disruption.
Also, because night workers and shift workers have difficulty sleeping during the day, we included sleep restriction along with that circadian disruption. So our circadian clock has an alerting signal during the daytime and a sleep signal at night, helping us to be awake and asleep at the usual time. So, most night workers sleep a couple of hours less during the day because of both the endogenously generated rhythm of alertness and sleepiness, and also because during the daytime, there's bright light, more noise, and the phone is always on.
Hannah - So they elongated the circadian rhythm and decreased the amount of sleep that the volunteers were allowed, effectively giving them a harsh version of rotational nightshift work.
Your body receives circadian cues which regulate how you respond to food. Muck about with your circadian clock and you muck about with your metabolism. Your body has evolved to eat during the day and not at night, with dozens of genes and enzymes involved in digestion and metabolism highly expressed during the day. Eat in the middle of the night and these enzymes are not readily available to digest the food and your body is confused. It can't process information to tell you when you're feeling full. Your satiety system is sleepy at night and it can't send the proper signals telling you that you've eaten enough.
Orfeu - We're very interested in what we call ecologically valid assessments of metabolism. So, we used the response to a meal. Everybody has to eat after all. And when you give a controlled and identical meal, we saw that when participants had circadian disruption, their glucose levels go very high and stay high for longer.
Hannah - And is that comparable to having diabetes in some way then?
Orfeu - Well three of our participants out of 21 achieved glucose levels 2 hours after the start of the meal that would meet the clinical criteria for pre-diabetes when they weren't that way at baseline. Fortunately, they recovered after 9 days of sleep at the normal circadian time. When we take in food, our pancreas responds by secreting insulin which promotes the storage of that glucose, and the use of that glucose in peripheral tissues, such as muscle. We saw a 27% decrease in the amount of insulin to the exact same meal when subjects were circadian disrupted.
Hannah - What kind of impact would this have in terms of weight gain?
Orfeu - Our exposure to circadian disruption increased obesity risk is via the change in another parameter called resting metabolic rate. That's our basal metabolism, subtracting out voluntary activity like exercise and so forth. So it's the amount of calories our body uses, just in the normal course of things without the addition of exercise. What we saw was an 8% decrease and while this number sounds small, it's sufficient that if someone had this 8% decrease in basal metabolic rate, and no change in diet or activity levels, they would put on 10 to 12 pounds in a year, taking a normal weight person to obesity within several years.
Hannah - So your results are showing a clear alteration in metabolism and also, glucose levels in those people that have altered circadian rhythms and diminished sleep. What can shift workers do about this?
Orfeu - Night workers have several options. The first is to better adapt their circadian rhythms to the imposed work schedule. So for example, by appropriately timing light and dark, and meals, it's possible to synchronise your circadian rhythms and be eating at a time when your body is prepared for the infusion of that fuel. In addition to adapting your circadian rhythms, the other key strategy is to allow time for recovery. There's chronic sleep restriction associated with night work because the circadian drive for alerting during the day makes it very hard to sleep. So the idea would be to sleep in a silent, dark room with blackout shades, and a cool room.
Ben - Harvard's Orfeu Buxton speaking with Hannah Critchlow. This study he was describing was published very recently in the journal Science Translational Medicine.
17:08 - Rod transplants restore sight to blind mice
Rod transplants restore sight to blind mice
Rod precursor cells transplanted from the eyes of newborn mice into blind adult animals can restore vision, UK scientists have shown.
Working at UCL and publishing their results in Nature, researcher  Robin Ali and his team used adult mice lacking a gene called Gnat1, which prevents the animals' rod cells from working, triggering night blindness. Into a group of these animals the scientists injected beneath their retinae 200,000 developing rod cells collected from 4-8 day-old donor mice.
Robin Ali and his team used adult mice lacking a gene called Gnat1, which prevents the animals' rod cells from working, triggering night blindness. Into a group of these animals the scientists injected beneath their retinae 200,000 developing rod cells collected from 4-8 day-old donor mice.
These donor cells expressed a genetic label to enable them to be distinguished from the recipients' cells. The result, three weeks later, was up to 26,000 new rod cells in each recipient retina, with most of the new rods clustered around the injection site.
Examined under the microscope, the new rods appeared to have wired themselves into the host retinae correctly and responded appropriately to flashes of light. Compared with control animals that showed no responses, the visual areas of the treated animals' brains also altered their activity in response to light stimuli and the animals could follow moving light spots with their eyes, proving that signals from the grafted rod cells were being appropriately transmitted into the nervous system.
The treated mice were also able 70% of the time to locate a hidden platform in a water maze under low-light conditions. Control animals trying the same task performed no better than chance.
According to the scientists, "the results demonstrate for the first time that transplanted rod-photoreceptor precursors can integrate into a dysfunctional adult retina and, by directly connecting with the host retinal circuitry, truly improve vision."
Some commentators remain sceptical, however, criticising the the work on the grounds that the Gnat-1 mutant mice used still have functioning cones in their retinae and also have rods that could be rendered functional by the injected precursor cells.

21:09 - Getting to Grips with Treating Paralysis
Getting to Grips with Treating Paralysis
With over 130,000 people suffering some paralysis as a result of spinal injury each year, devices that can bypass the spine and convert brain activity directly into coordinated movement could immeasurably improve thousands of lives. Now, research at the University of Pittsberg and Northwestern University has done just that - returning the ability to grip a ball to partially paralysed monkeys.
Functional Electrical Stimulation, or FES, has been extensively studied and deployed as a way to restore movement to paralysed limbs. The technique involves using electrodes to simulate the signals from the brain and stimulate muscles directly. Clinical systems already exist that can return limited gripping ability to patients, but these generally involve tapping into the activity of a nearby healthy muscle, and learning to use that to control the hand in a simple pinch motion. Returning the full use of the hands would be life changing.
 Writing in Nature, Lee Miller and colleagues describe how they measured the electrical activity in the hand region of the primary motor cortex of monkeys as they completed some simple tasks. Importantly, they also measured the electrical activity in the muscles that controlled the hand and forearm, allowing them to link the brain and muscle activity and therefore predict how the muscles should move in response to electrical signals in the brain.
Writing in Nature, Lee Miller and colleagues describe how they measured the electrical activity in the hand region of the primary motor cortex of monkeys as they completed some simple tasks. Importantly, they also measured the electrical activity in the muscles that controlled the hand and forearm, allowing them to link the brain and muscle activity and therefore predict how the muscles should move in response to electrical signals in the brain.
The monkeys' two tasks were simple, but needed some fine control of grip. In one task, the monkeys had to pick up a ball roughly the size and weight of an apple, and drop it into a collecting tube to get a reward. The second task measured control of how hard the monkey was gripping, and involved squeezing a flexible tube with a particular strength for a given length of time.
Once the monkeys were trained, and their brain and muscle activity measured, the symptoms of a spinal injury were temporarily mimicked by knocking out the nerves in the elbow with anaesthetic. The researchers then used functional electrical stimulation along with their predictions of the response to brain activity in order to activate the muscles in real time.This allowed the monkeys to complete both tasks again, showing that this technique can return not just a binary on/off gripping motion, but also the fine control needed to modulate grip strength. This effectively bypassed the spinal cord completely.
Knocked-out nerves are a simplistic model of spinal injury, so this will take some development before we see it in use in human patients, but this is a major step forward and proof of principle for brain-controlled functional electrical stimulation.
24:37 - Reaching Further with RFID
Reaching Further with RFID
with Dr Sithamparanathan Sabesan, Cambridge University
Chris - If you've ever walked out from a shop and set off an alarm - I'm sure you haven't for anything other than accidental reasons - then you've probably triggered an RFID, that's Radio Frequency ID tag system. The problem with these tags is that although they can identify an individual piece of merchandise and are therefore very useful, they only work over very short distances and that limits their use.
But now, new research from Cambridge University has found a way to dramatically increase this range and therefore, the usefulness of these pieces of equipment. To tell us more, Sithamparanathan Sabesan who is from Cambridge University's department of engineering where he's a research fellow, is with us to tell us how it works. First of all, thanks for joining us. What do these RFID tags actually do? How do they do what they do?
Sithamparanathan - RFID stands for Radio Frequency Identification. These RFID tags do not have an internal power source, you have to power them up for them to receive signal and retransmit signal for example. And so, we have antennas. They transmit an electromagnetic signal for them to basically receive the signal. And these RFID tags then have a unique ID stored in the microchip.
Chris - So, the energy comes in from a radio signal from a transmitter. It interacts with the tag which gets its energy from that, powers up a little microchip which sends a unique radio signal back to a detector and that says, "Hi. Here I am" and identifies that item uniquely.
Sithamparanathan - Absolutely.
 Chris - So that sounds terrific. What is the problem with these tags? Why are they an issue?
Chris - So that sounds terrific. What is the problem with these tags? Why are they an issue?
Sithamparanathan - The real challenge here is then, because they don't have a battery, it's very difficult for us to read this tag over longer distance, due to the fact that the signal not only travels in one single direction, but also, travels in multiple directions.
Chris - Because of in reflections off of walls and the floor, and so on.
Sithamparanathan - Exactly. So the results of that, the direct signal gets cancelled out by the reflected signals.
Chris - So this is the signal coming in from the transmitter that you're beaming to the tags. It will bounce off all the surfaces and in certain places, you're going to cancel out the signal and get a dead spot.
Sithamparanathan - Exactly and we call them dead spot. The reality is that, over a large area, you'll have so many dead spot that typically, you can only read 60% of the tags in a conventional approach. And so, we developed a technology called dithering. We have multiple transmitters, they synchronise in a way that we are able to move this dead spot around within that area. As a result of that, I shall be able to read this tag with 100% accuracy. I won't miss any single tag over that large area.
Chris - Wow! So, where previously with a static array of transmitters, you would end up with a pattern of dead spots. It's a bit like if you walk around your house with a mobile phone, it works in some rooms and not others. You've got a pattern of dead spots and so, if the tag is in one of those, you're not going to spot it. But with your system, you're moving the dead spots around in the environment, dithering them. So, even if a tag is in a dead spot one moment, the next moment it's not and you're going to get the signal back. And so, the trick is how you move the dead spots then?
Sithamparanathan - So we use multiple transmitters in a synchronised way by doing some clever advance signal processing at the backend and also synchronise them. These are able to move these dead spot very quickly because you got to move them very fast. It's typically in milliseconds kind of speed. By doing so, over the 1 second period, I shall able to read all the single tags.
Chris - And this means that then, rather than just having to put in massively powerful transmitters which still wouldn't get around the dead spot problem, you've now got a simple array, you use these very cheap tags and you can detect them over, I would say, probably much greater distances as well then.
Sithamparanathan - Yes. So far, we have demonstrated over 20-meter x 20-meter area with 100% accuracy. You could scale that to a large building, typically hundreds and hundreds of meter large buildings, you could have a small number of antennas and read all the tags over that area.
Chris - How do you actually read where the tags are? In order to scale it over a very big area like that, you're detecting a tiny signal coming out from a tag which could be anywhere in that environment? So is that a sort of triangulation trick then?
Sithamparanathan - Exactly. So, in order to locate the tags, that's another challenge. Reading a tag is one challenge we just discussed and locating the tags is another challenge. You could have triangulation. Even then, you're still going to have the multipath and dead spot problem, but we have another invention which enable us to triangulate these tags in a novel way. The results of that is that you can locate these tags within 1 meter location accuracy.
Chris - So supermarkets are going to be delighted because I'm thinking, all I need to do is I go into the supermarket, swipe my credit card and identify myself electronically to the supermarket with a sort of trolley or something and then every time I take something off the shelf, the supermarket knows where the object is because you can have these tags on every piece of stock. So a.) It's good for stock control, b.) It stops shoplifting, and c.) It means that I don't have to queue up and check out. I just walk out.
Sithamparanathan - Exactly, so supermarket automatic checkout is one of the long term visionary application we're focusing on. As you said, in the future, you don't need to stand in a queue. You just walk with your trolley and pass through that reader and it will give you the bill straight away, there's no queue whatsoever. But there are other applications we're currently launching, one of which is document tracking. Working with local councils where they want to see where every single file is within large office area...
Chris - I think also in a hospital because tracking down a patient's notes - it sounds trivial but if you're in the operating theatre and the notes are on the other side of the hospital, knowing where they are to send someone to get them could be really helpful.
Sithamparanathan - I'm glad that you mentioned hospitals because that's one of the customers we've been speaking with very recently. They will also want to track their patients because they also want to learn how their diseases spread within hospitals.
Chris - Have you got any trial data to show that this will actually work though? It's one thing to say, "Right, we think we can do this in supermarkets, we think we can make this work in a hospital to track people and notes." What about the reality though? Have you got data on this?
Sithamparanathan - So very recently, we have developed and demonstrated for the first time a document tracking system. We were able to show that we were reading hundreds of files over that large area. We were demonstrating over 20-meter x 20-meter area. There's also another big launch that happened in London last week, for tracking containers and cars in over a large area and so, so far, we have a prototype system which is able to show that we have 100% coverage over that large area for the first time.
Chris - Amazing stuff, thank you very much. That's Sithamparanathan Sabesan, from the Department of Engineering at Cambridge University.
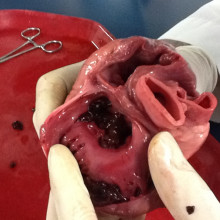
31:23 - Scar tissue re-programmed to become beating heart cells
Scar tissue re-programmed to become beating heart cells
Scar tissue in injured hearts can be converted back into healthy cardiac muscle using gene therapy, US scientists have shown.
Following a heart attack, some of the muscle tissue that 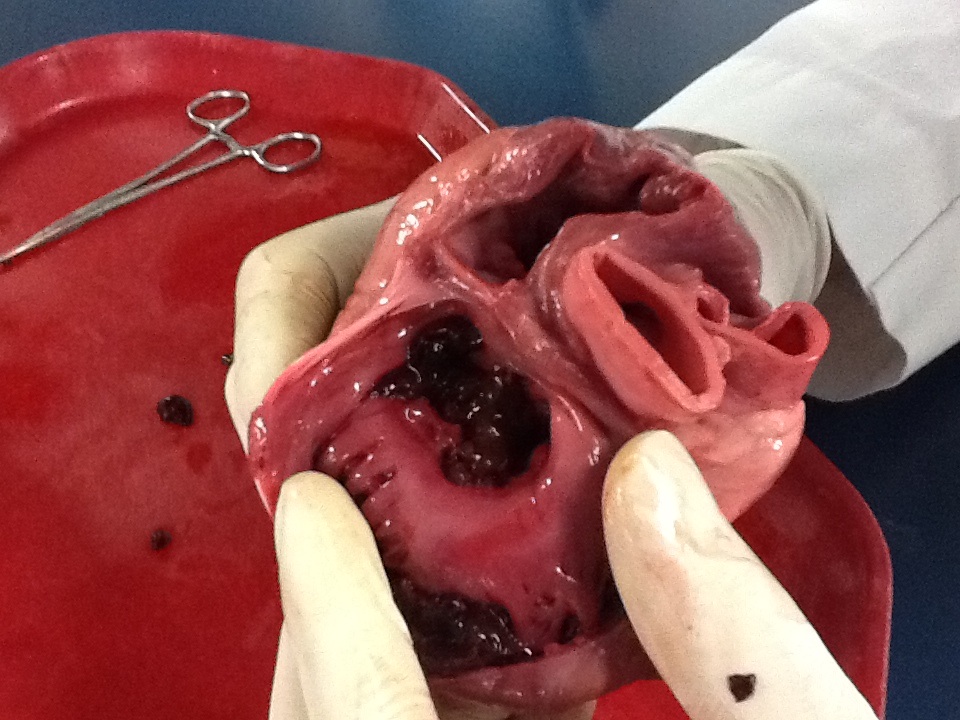 generates the pumping action needed to push blood around the body is replaced by an immobile fibrous scar.
generates the pumping action needed to push blood around the body is replaced by an immobile fibrous scar.
If it's sufficiently extensive this can compromise the function of the heart, causing heart failure. The scarred areas may also affect the way that the electrical signals that coordinate the heartbeat travel through the tissue, triggering abnormal rhythms and even cardiac arrest. And because heart muscle doesn't naturally replace itself after an injury, these consequences were thought to be permanent.
Now, writing in Nature, Deepak Srivastava and his colleagues at the Gladstone Institute of Cardiovascular Disease in San Francisco may have found a way to convert the fibroblast cells that form scar tissue back into beating heart cells, in-situ.
The discovery hinges on three genes called Gata4, Mef2c and Tbx5; these are known as "GMT" and have been shown previously to operate during the embryonic development of the heart to direct the formation of heart muscle cells known as cardiomyocytes.
By engineering these genes into disabled viruses, which were then injected into the damage parts of the hearts of experimental mice that had had recent heart attacks, the team successfully reduced the sizes of the scars that subsequently formed in the animals' hearts and demonstrated that, compared with controls, the animals had improved cardiac function as a result.
A dose of about 100 million virus particles was able to hit about 300,000 cells, converting about 12% of them into new cardiomyocytes. And because the technique labelled-up the newly-re-programmed cells, the scientists were able to confirm their origins and test their function.
The cells wired themselves apropriately to adjacent cells and contracted when electrically activated. They also had the correct appearance, genetic and biochemical profiles.
In their paper the team conclude, "The ability to regenerate adult heart tissue from endogenous cells is a promising approach to treating cardiac disease that may face fewer obstacles to clinical translation than other approaches..."

33:41 - Water Sources in Africa and Orangutan Engineers
Water Sources in Africa and Orangutan Engineers
with Helen Bonsor, British Geological Survey; Graham Hardie, University of Dundee; Bernard Kippelen from the Georgia Institute of Technology; Roland Ennor, University of Manchester
New Water Source Revealed throughout Africa
Water availability across Africa could be improved significantly by tapping into reservoirs of groundwater.
Writing in the journal Environmental Research Letters, Helen Bonsor from the British Geological Survey collated data from regional maps and scientific literature and mapped the potential yield of groundwater across the African continent. The findings estimated the total volume available to be 0.66 million km2, more than 100 times that available on the surface annually. However, levels were found to vary greatly in different regions.
Helen - The amount of groundwater available in Africa varies considerably and it's not available for widespread development of high yielding bore holes across the continent. There's only large volumes of groundwater stored in North Africa, so in Libya, Chad, Algeria. In Sub-Saharan Africa, where most of the population live, there's actually much lower groundwater volume stored there. If it's exploited very carefully then it is possible that groundwater could support small scale drinking water supplies where there are 300 million people who don't have basic access to water.
New Disease Targets for Aspirin
A new effect of aspirin in the human body has been discovered by scientists at the University of Dundee. Once consumed, aspirin is rapidly broken down into the compound salicylate. When applied to human cells in the lab, Graham Hardie found that high concentrations of this compound activated the enzyme AMPK - a regulator of cell growth and metabolism. It's thought this effect of aspirin, away from its pain-killing properties, could be beneficial against a range of diseases, including cancer...
Graham - It's been found that the people who take aspirin long term tend to have a lower incidence of cancer. So, it's at least possible that the effects of taking regular aspirin on cancer might not be mediated by the sort of classical target, but might be mediated instead by this new target which is AMPK. We haven't proved that yet, but it raises an exciting prospect that this very old drug might actually have some new applications.
Polymer for cheaper solar cells
A new polymer could enable cheaper, easy to produce solar cells. To be able to generate electricity, organic electronics, such as solar cells, require the use of materials that receive and release electrons readily. This usually means the use of conductors such as calcium or magnesium which are highly reactive with air and water and as a result are coated with thick, expensive materials to stabilise them. But now, a new polymer developed by Bernard Kippelen from the Georgia Institute of Technology has been shown to achieve this property in less reactive materials when applied to them as a thin coating...
Bernard - These polymers are air stable, so they don't degrade in contact with air, and we hope that the use of air stable materials in the field of organic photovoltaics will accelerate the deployment of these technologies through reduced cost and simpler manufacturing.

Orangutan Engineers
And finally, orangutans use advanced engineering skills when building their nests, according to scientists at the University of Manchester. By observing 14 orangutan nests in Sumatra, the team found that strength, flexibility and comfort were taken into account by the apes with different branch types being used for various aspects of design. Thick branches were only half split and weaved together to make a strong basket-like structure, and thinner branches used to construct a comfortable lining to sleep on. Roland Ennos is a senior member of the research team.
Ronald - Orangutans are much more sophisticated in their use of branches in producing nests than we hitherto have thought. They seem to have some knowledge of the properties of the wood and how it will break, and they make best use of those properties. And this has implications for the evolution of intelligence. It suggests that we are not the only toolmakers and that the evolution of intelligence might be related to the ability of organisms to manipulate their mechanical environment, not just their social environment.
The work was published in the journal PNAS.

38:21 - Frog Fungi - A Global Problem Affecting Amphibians
Frog Fungi - A Global Problem Affecting Amphibians
with Trent Garner, Institute of Zoology & and Matthew Fisher, Department of Infectious Disease Epidemiology, Imperial College London
Chris - Amphibian species around the world are subject to an increasing threat in the form of a fungus. Over 200 amphibian species are thought to have become extinct and the problem isn't just restricted to frogs, toads and newts. During the 19th century, for instance, a fungal infection caused the potato famine in Ireland and, according to new research, it could affect food security today.
Planet Earth podcast presenter Sue Nelson went to London Zoo's reptile house - of Harry Potter fame - to meet up with Trent Garner, from the Institute of Zoology, and Matthew Fisher from the department of infectious disease epidemiology at Imperial College London.
She began by asking Matthew why we haven't we heard more about fungal infections affecting biodiversity...
 Matthew - Fungal infections haven't really been a problem until the last two decades and what we've noticed is that there are far more infections now than there used to be. I mean we know about fungi driving frogs extinct but there have also been very aggressive emergences in bat populations in North America, bat white-nose syndrome, and we're also seeing dramatic emergences in forests, so we all know about Dutch Elm disease but also in crops too. There are new virulent forms of fungal lineages which can devastate entire crops, so UG99 has a demonstrated potential to wipe out a quarter of the world's wheat supply. So, we've been thinking very hard about why this is occurring and what these trends that we're observing actually mean.
Matthew - Fungal infections haven't really been a problem until the last two decades and what we've noticed is that there are far more infections now than there used to be. I mean we know about fungi driving frogs extinct but there have also been very aggressive emergences in bat populations in North America, bat white-nose syndrome, and we're also seeing dramatic emergences in forests, so we all know about Dutch Elm disease but also in crops too. There are new virulent forms of fungal lineages which can devastate entire crops, so UG99 has a demonstrated potential to wipe out a quarter of the world's wheat supply. So, we've been thinking very hard about why this is occurring and what these trends that we're observing actually mean.
Sue - Let's look, in particular, at how a fungus affects one particular species and that's frogs. Trent, how bad have frog populations been affected?
Trent - In some parts of the world they are thinking that species have actually gone extinct due to this fungus and that's a rather unusual thing, to have a parasite drive its host to extinction. Not only has this happened in one part of the world but it has actually happened on multiple continents and that's the thing that's so worrying that such an unusual occurrence can actually be replicated on a global scale. Of course that's not just species extinctions that are happening, we're seeing catastrophic population declines where the remnant populations are so small that they could go extinct at the blink of an eye for any reason, because there's just not enough numbers there to maintain the population and again this is occurring at a global scale.
Sue - I think it's time, listening to those helicopters, to actually go inside the reptile house, get out of the noise here, and perhaps go and see some of the frogs that have been affected...
...It's still noisy but it's a popular time, lots of school children and familiar glass fronted cases containing snakes and lizards, geckos on my left there, African Bull Frog and a Blue Poisoned Dart Frog - oh, that is beautiful, that is a vivid blue isn't it?
Trent - They are beautiful aren't they? They come from a family of frogs that has been heavily affected by the fungus in Latin America. Some of the species within the family are presumed extinct due to the emerging infectious disease.
Sue - Is the fungus that affects these frogs the same, Matt, that affects all frogs around the world or are there different strains?
Matthew - No, there's one single strain that's emerged and we've dated that to the mid 1970s or perhaps a little bit earlier than that and that seems to have emerged pretty much simultaneously in five continents.
Trent - If we go over here there's actually a salamander species called the axolotl and it's a member of the family of ambystomatid salamanders and, while we don't have any evidence of this species actually being affected by the disease, they certainly are infected with the fungus, and it's a critically endangered species.
Sue - Matthew mentioned about the spread over five continents, what has caused the fungus to be spread in that way?
Trent - I think that's a really good question and I think we're still developing the evidence base to actually answer that question. Certainly, a lot of people invoke amphibian trade as being responsible for the spread and we do have some evidence that amphibian trade has been responsible for some spread events. Exactly how it's been responsible for spread overall is yet to be determined.
Sue - When you have a fungus that can affect and potentially devastate crops of wheat or rice that's extremely worrying. What can be done about this?
Matthew - We would argue that much stronger international bio-security is necessary. We see the doors to our country as absolutely wide open for the trade of animals and plants in the nursery trade or in the pet trade and these hosts have the demonstrated potential to carry new infections into countries and we're witnessing the effect of that, the manifested potential of these pathogens. So we need to quarantine more and we need to give the international organisations which control trade in bioactive material more muscle.
Sue - Do you think it's too late, Trent?
Trent - No. I agree with Matt that we need to tighten up regulations and tighten up enforcement to reduce the risk of disease being transported around. But even if disease occurs in an area, even if disease does emerge in an area, I do think that steps can be taken to reduce forcing of infection and potentially reduce the effects of the parasite without necessarily eliminating the parasite.
Chris - Trent Garner from the Institute of Zoology and Matthew Fisher from the department of infectious disease epidemiology at Imperial College London. And a longer version of that report from Sue Nelson can be heard in the latest Planet Earth podcast. You can download it from our website or via Planet Earth Online.
44:06 - How Protein Pathways Set Cell Clocks
How Protein Pathways Set Cell Clocks
with Dr. Akhilesh Reddy, Institute of Metabolic Sciences, Cambridge University
Ben - Returning to our topic of circadian rhythms, the body clock and sleep, it seems that circadian rhythms are clearly important for staying fit, both in terms of good health and in terms of survival of the fittest. They fit chemical cycles to the day and night pattern, and this seems to have offered an evolutionary advantage. But why would that be and what are these essential chemical pathways that encouraged body clocks to evolve?
To find out more, we are joined by Cambridge University's Ak Reddy. Ak, thank you ever so much for joining us. First off, what do we think the evolutionary advantages are of having a body clock?
Ak - Well, the ideas that have been bandied around in the past are to do with trying to separate out things that should happen in the day and night, and one of the great examples that was used in the past was about cell division cycles. So, in the days when cells were first evolving, people thought that it might be advantageous to protect DNA replication which is very sensitive to UV radiation from the Sun and have that going on in one part of the day and then not have that protection at night time for example. Then you separate out these two phases - hHow do you do that? You have a timing mechanism that basically keeps track of when the day and night is going to come, and basically wire it into the way that the cell divides; the cell cycle.
 Ben - So, do we see it in a wide range of species? We've already discussed the fact that plants seem to show a similar circadian rhythm as well..
Ben - So, do we see it in a wide range of species? We've already discussed the fact that plants seem to show a similar circadian rhythm as well..
Ak - Absolutely. In fact, animals and plants dominated how we thought about clocks, and it wasn't until about 20 years ago that people first started looking in bacteria. People had mentioned bacteria or mentioned the fact that bacteria may have clocks many years ago, but it kind of got wiped out because people thought, well why does a bacterium, something that divides every half an hour, or even more rapidly in some cases, why does that need to have any knowledge of a 24-hour cycle? But it turns out actually that bacteria did evolve to basically do what I said before and protect themselves from DNA damage in parts of the day versus the night time and so, they actually prefer dividing in the night time compared to the day time.
Ben - So, even though you'll have multiple generations in the space of a 24-hour cycle, you will still see a circadian rhythm.
Ak - Exactly. So they divide. They continue on dividing, but they prefer to divide in the night time and we know that that happens in ourselves as well, in something that divides regularly like the liver for example.
Ben - So what's the chemical background? Is it genetically controlled or is there essentially a simpler process?
Ak - Well it's actually a combination of both things. So in a complex cell like our cells, we think that there are two main ways in which the "clockwork" is made up. This is what we currently know. The one that we talked about before was genetics, which is switching things on and off, basically, over a 24-hour cycle. What we've discovered recently is that you can also get cycles without any genes whatsoever. So you make proteins, they need to be there, you need a blueprint if you like, which is the DNA genetic code. But once you make your proteins, they can actually oscillate on their own without genes being switched on and off, and we actually showed that in red blood cells, which don't have any DNA, and they don't make any new protein, but they're still able to tick and tock over the 24-hour cycle without any influence from the outside world.
Ben - Speaking of influence from the outside world, what happens if you take away all of the external stimulus? What about the bacteria that are at the very deepest deep seas that never see any daylight at all and those sorts of species? Do they keep going?
Ak - They keep going, yes. As far as we know, I mean, we haven't gone to the depths of the ocean and picked out those bacteria, but people have worked on cave fish for example, that have been kept isolated for many, many generations to the extent that they even lost their eyes, so they don't actually bother keeping track of light anymore, but they still seem to maintain pretty ropey but present circadian rhythms.
Ben - I guess there are other things that fit the same pattern, so even if you're not seeing any light, there will be changes in temperature perhaps, changes in tides, the way the water moves, and so on. Do we think that these other physical factors might help keep things in check?
Ak - Absolutely. Temperature is probably one of the most fundamental synchronisation cues. So we tend to think that light is the dominant thing, but temperature is something that's also been present ever since day one in terms of an oscillation, and indeed, oxygen cycles as well which is, is again, another synchronisation stimulus, and feeding cycles as well. So, food availability from up above, stuff at the top of the ocean may feed down to stuff more deep in the ocean. So everything is cyclical in the ocean and there are even tidal rhythms as well that overlap onto this.
Ben - So, now that we have a better idea of what this protein clockwork pathway really is, can that tell us why, or can that give us some good avenues to look out for why it is that disrupted circadian rhythms seem to have these deleterious health effects?
Ak - With all kinds of science and medicine, we need to really kind of understand the basics before we can start applying it and I think we're at the stage now where we have enough information about all of the different components of the molecular clockwork and we can actually start to kind of unravel how they might go wrong in diseases. There've been a few diseases associated with mutations in particular circadian rhythm genes, but not that many. Maybe we've been looking in the wrong places or we're just not looking at the right genes.
Ben - So there's lots of new avenues for research, just from our basic understanding.
Ak - We hope so.
Ben - Well, thank you very much. That's Ak Reddy. He's from the Institute of Metabolic Science here at the University of Cambridge.
Is nausea related to sleeplessness resulting from blindness?
Russell - What we know is that if you have no eyes, then your ability to regulate internal time is effectively lost in humans and so what Angie will be experiencing is essentially a constant drifting of the body clock. Now, Allan Bird at the Institute of Ophthalmology has been able to stabilise individuals with no eyes by using 3 milligrams of melatonin in the evening. And so, what will be useful is either Angie can contact her local GP and she needs to be advised by an ophthalmologist.
Can stress lead to circadian disruption?
Russell - Stress is probably one of the ways in which shift workers for example are suffering many of the pathologies. One thing we know, for example, is that elevated stress will elevate cortisol, will reduce the immune system, and of course, levels of infection are much higher in shift workers compared to day shift workers. So stress is going to be really important in our bodily responses to try and function when the clock is not in sync with the environment.
Why would it matter to bacteria when they divide?
In our interview with Ak Reddy, he explains how species segregate certain tasks into day or night - DNA replication, as needed for cell division, is affected by UV radiation from the Sun, leading to mutations. As a result, many species prefer to do the majority of cell division at night...
Ak - In terms of bacteria, it obviously doesn't think about things, it just tries to grow the best it can essentially. But essentially, if it wants to survive, its survival of the fittest, it wants to make sure that its progeny basically are the ones that live on and capable of living on to the best that they can then they need to basically make sure that they survive and don't accumulate mutations that they don't actually need.
Can your skin sense enough light to help with timing issues?
Ak - I think the bottom line is that skin cells, although they have intrinsic circadian clocks within them, cannot receive the light and make use of that light. They need the Suprachiasmatic Nucleus (SCN), the master clock in the brain to actually be able to talk to those cells and resynchronise them.
How do Mars scientists cope with the circadian shift?
Russell - The Martian day is 24 hours and 36 minutes and of course, the Mars day is not going to map onto the day a lot of the time in Houston. In fact, when the Martian rover was trundling around, it was working brilliantly of course during the Martian day, but sometimes it was out of sync with mission control. The mission control guys got so tired, because it was out of sync, that they turned the rover off because they couldn't function.
Are ganglion cells more sensitive to certain colours?
Russell - We've shown in both mice and humans that it's at 480nm, the blue part of the spectrum. You can imagine it as a bell-shaped curve with a peak being at 480nm in the blue part of the spectrum. But it's worth saying that we originally discovered these cells because we were looking at how the clock is regulated by light. But actually, what these cells are doing is plugged into a whole raft of different brain structures in the hypothalamus and the thalamus, and they're regulating lots of brightness detection tasks generally. These are not just clock regulators. So for example, part of our pupil constriction in response to bright light is via these photosensitive ganglion cells, again, blue light sensitive. Part of our arousal systems and part of our sleep systems are all being regulated by these extraordinary cells.
Ben - Different types of light bulb give out different ranges of light. What about when we're using energy saving light bulbs which give out these very distinct bands of radiation? If that doesn't happen to coincide with the blue that we need to set our day, then does that mean that all of these energy saving light bulbs might actually be disrupting us?
Russell - Well it raises a really very interesting issue because what you want to do is fine-tune artificial lighting systems to enhance reading at certain times, but also, you want to either stimulate these photosensitive ganglion cells, increase alertness let's say, or not stimulate them at night when you want to go to sleep. So, there's increasing attention being paid to fine-tune artificial light sources to stimulate both the visual system versus this photosensitive ganglion cells.

54:56 - Would reading from a screen keep me awake?
Would reading from a screen keep me awake?
We put this question to Professor Debra Skene from the University of Surrey:
Debra - I do studies on light and sleep. We know that light can affect alertness and it can affect your ability to sleep. We know that it in fact stimulates receptors in the eye that are particularly sensitive to blue light. This light information then is sent all around the brain and can affect how tired you are and possibly, your ability to sleep. So I would say, if you had very bright light in your bedroom and we know this from studies, that it in fact delays your sleep time and keeps you more awake. Now about electronic books, they're not very bright in terms of the intensity of light and so, I would suspect that the light levels are too low to really affect your sleep. If you wanted to use a computer that had a very bright blue screen, this may keep you up.
Hannah - Computer screens, televisions and bright lights all emit a strong blue light of high enough intensity to stimulate. So, reading from a backlit screen of a laptop or tablet could lead to sleepless nights. However, most E-Readers use special electronic paper technology which has no backlighting and utilises reflected light in exactly the same way as normal paper. Because of this, an E-Reader should only keep you awake if you can't put it down.










Comments
Add a comment