Join us to explore the virus behind Hepatitis C. We'lle be investigating the causes, prevention and treatment of this often masked but serious disease.
In this episode

- How many classes of hepatitis virus are there and how are they different?
How many classes of hepatitis virus are there and how are they different?
We put this to Graeme Alexander:Graeme - Well a lot of viruses can affect the liver, but there's five that we recognize as important in the liver and they're HepatitisA, B, C, D, and E - rather imaginative aren't we?The most important thing about hepatitis C is that it, by and large causes an infection that lasts for a lifetime while the other viruses don't (usually). So that differentiates it quite easily. And it's an RNA virus whereas hepatitis B is a DNA virus infection. But that's sort of a semantic difference. The important thing about hepatitis C is that it causes lifelong infection in many people.

New way to lock up carbon dioxide
Scientists may have found a much more cost-effective way to extract the CO2 from exhaust gases.
 With Copenhagen just around the corner the attention of the world is firmly fixed on the question of cleaning up our emissions. But efficient ways to selectively scrub CO2 from the waste gas streams leaving power-stations or other heavy industries have proved hard to find.
With Copenhagen just around the corner the attention of the world is firmly fixed on the question of cleaning up our emissions. But efficient ways to selectively scrub CO2 from the waste gas streams leaving power-stations or other heavy industries have proved hard to find.
The best contenders to date have involved dissolving the gas in solutions of amines, but this is problematic because to re-release the gas from the amine - to store or sequester it - consumes large amounts of energy, making the process inviable in energy terms.
But now a new molecular structure may have come to the rescue, the MOF or metal organic framework. These are large molecular structures resembling chemical cages linked together in a repetitive sequence. But what's special about these cages is that the 'bars' consist of organic molecules that plug into metal atoms at the vertices and this combination gives these structures very exciting chemical properties including the ability to selectively lock away certain gases inside the cages but simultaneously reject others. This means that they can behave like molecular sieves and California Nanosystems Institute scientist David Britt and his colleagues have developed a MOF that is very good at selectively grabbing CO2.
Writing in PNAS, the team present a molecule called Mg-MOF-74, which contains magnesium atoms linked to the organic material DOT, short for 2,5-dioidoterephthalate. Exposed to a mixture of gases that includes a low concentration of CO2, this MOF selectively sequesters the carbon dioxide by linking it to the magnesium atoms inside the molecular cages; the other gas molecules meanwhile, slip through unimpeded. Fully charged the MOF can soak up close to 9% of its weight in CO2; better still is can re-release the CO2 just with gentle warming to only 80 degrees C, liberating the gas and regenerating the MOF.
This say the scientists meakes these molecules excellent candidates as CO2 capture media; they also offer the advantage over existing methods of being non-toxic and non-corrosive, which are unpleasant features of the amine solutions currently being trialled.
Progress in cystic fibrosis
Researchers in California have discovered a way to partially repair damaged lung cells from patients with cystic fibrosis, an inherited disease that affects more than 70,000 people around the world. The results are published in the journal Nature Chemical Biology this week, led by Professor William Balch and his team at the Scripps Research Institute.
 It all centres on protein processing - when proteins are made in our cells, they are folded up into the correct shape. But if there's a fault in the protein, or in the folding process, then the protein doesn't work properly and it gets broken down again.
It all centres on protein processing - when proteins are made in our cells, they are folded up into the correct shape. But if there's a fault in the protein, or in the folding process, then the protein doesn't work properly and it gets broken down again.
In cystic fibrosis, the disease is caused by faults in the gene that makes a protein called CFTR, which normally sits on the surface of cells and helps to shuttle salts across the cell membrane. Around nine out of ten people with the disease have a faulty version of the CFTR known as DF508 CFTR. The resulting protein is the wrong shape, and gets broken down in the cell's endoplasmic reticulum - the molecular factory where proteins are made.
The researchers figured that if they could stop the DF508 version of the protein being degraded, then it might work at least a bit, and help to relieve some of the symptoms of cystic fibrosis - and that's just what they've managed to do.
The scientists used a drug called suberoylanilide hydroxamic acid, or SAHA for short, which blocks enzymes called histone deacetylases. These normally work to affect the proteins that package DNA in the nucleus of the cell, helping to switch genes on and off, but they also have other effects on the processing of proteins.
They tested the drug on lung cells taken from cystic fibrosis patients with the DF508 fault, and found that SAHA treatment restored the level of CFTR activity to 28 per cent of that found in normal lung cells.
That may not sound like a lot, but it could make a real difference. For example, patients with less severe cystic fibrosis, with around 15 to 30 per cent levels of CFTR activity, can lead a much more normal lifestyle, compared with people carrying a more severe fault like DF508. So being able to restore 28 per cent of lung cell function could really be significant.
The scientists also found that SAHA works best at relatively low doses, which is important if it's to be taken forward for clinical trials. The other good thing about the drug is that it has already been used in clinical trials for treating cancer, but in higher doses over a short period of time.
More research and tests need to be done to find out if it's suitable for giving in lower doses over a lifetime, which would be the case for treating people with cystic fibrosis. But these early results are certainly promising. And Balch thinks that a similar approach might also work for other conditions such as type II diabetes, arthritis, osteoporosis, and Alzheimer's disease.

Mobile phones off the hook as cause of brain cancer?
A large study of brain cancer cases has failed to find any increase in line with mobile phone use.
 Writing in the Journal of the National Cancer Institute, Danish Cancer Society scientist Isabelle Deltour and her colleagues looked at 60,000 patients with brain cancers diagnosed between 1974 and 2003 from a population of more than 16 million. The researchers found that the incidence of gliomas and meningiomas among adults was stable over this time and, critically, there was no change reflecting the sudden surge in mobile phone use that occurred in the mid 1990s.
Writing in the Journal of the National Cancer Institute, Danish Cancer Society scientist Isabelle Deltour and her colleagues looked at 60,000 patients with brain cancers diagnosed between 1974 and 2003 from a population of more than 16 million. The researchers found that the incidence of gliomas and meningiomas among adults was stable over this time and, critically, there was no change reflecting the sudden surge in mobile phone use that occurred in the mid 1990s.
The authors acknowledge that the time between mobile phone exposure and the development of a brain tumour could be greater than the 5-10 year lead-time investigated in the study, and they also point out that, if such an effect does exist, it may also be be too small to be observed here. But, more reassuringly, it could also confirm that there is no increased risk of brain cancer conferred by mobile phone use.
As we have no way of knowing which it is, for the meantime at least, we'll just need to hang on to find out...

Come and have a go if you think you're hard enough
Researchers from Caltech, the California Institute of Technology, have made a step forward in understanding how aggression may be hardwired into the genes, at least for fruit flies.
 This is research from Professor David Anderson and his colleagues, writing in the journal Nature this week. They've found a chemical pheromone that controls aggression in flies, and have also pinpointed the nerve cells in their antennae that detect the pheromones and send signals to the brain, telling it to kick off.
This is research from Professor David Anderson and his colleagues, writing in the journal Nature this week. They've found a chemical pheromone that controls aggression in flies, and have also pinpointed the nerve cells in their antennae that detect the pheromones and send signals to the brain, telling it to kick off.
We've known for a while that insects can respond aggressively to certain chemicals, or pheromones, when they're presented with artificial versions of them. But we don't know how much they use these pheromones normally to control their aggression. To prove it, the scientists had to track down the exact receptors in insect nerve cells that receive the pheromone signals - something that could only be done using fruit flies, as scientists have done a lot of research into their nervous system.
The researchers discovered that a chemical called 11-cis-vaccenylacetate, or cVA for short, can make pairs of male flies get aggressive, rearing up on their hind legs and hitting each other with their forelegs. And when they put pairs of male flies near a mesh cage containing other males that were releasing the chemical, they also became aggressive. But when the researchers silenced the nerve cells that respond to cVA, the flies no longer showed the aggressive behaviour.
Male fruit flies gather on food, because it gives them opportunities to mate with passing female flies. Normally they all get along OK, but if there are too many male flies, then this might interfere with feeding and mating.
The researchers took male flies that had been genetically manipulated to have hyper-sensitive nerve cells that detect cVA. They found that when these flies gathered on food, they fought each other until there was just one victor left. But when they tested unmodified flies, they just gathered together happily.
The researchers thinks that when the population of male flies gets high enough, the levels of cVA that they produce rises. This makes the flies aggressive and they fight, driving away some of them. As the flies fly off, the concentration of cVA drops again, and the flies calm down - and this cycle keeps repeating.
At the moment, these experiments have just been done in the lab, but the team thinks that it should be possible to reproduce them in the wild. And it would be very interesting to find out if the same kind of thing is at work in humans. Researchers have found aggressive pheromones in mice, so it's possible that we may also have them.
13:20 - Hitting Hepatitis C
Hitting Hepatitis C
with Henrik Øren, Santaris Pharma
Chris - Also in the news this week, in the journal Science, there's a paper which highlights a potential new treatment for hepatitis C. This new paper describes a molecule which will target hepatitis C by attacking a microRNA, a short piece of genetic material, which liver cells make and which seems to be absolutely critical for the virus to be able to replicate or grow. One of the people who's helped to make this possible is Dr. Henrik Øren from Santaris Pharma. He's with us now. Hello, Henrik.
Henrik - Hi.
Chris - Welcome to The Naked Scientists.
Henrik - Thank you.
Chris - So please tell us first of all, what is the problem with hepatitis C, actually treating it at the moment with existing therapy?
 Henrik - Well, at this time, there's probably about a couple of hundred million hepatitis C sufferers worldwide and the standard care is a combination of interferon and ribavirin which is effective in only about 50% of patients and associated with significant adverse effects.
Henrik - Well, at this time, there's probably about a couple of hundred million hepatitis C sufferers worldwide and the standard care is a combination of interferon and ribavirin which is effective in only about 50% of patients and associated with significant adverse effects.
Chris - So what you're saying is that we can't do much about hepatitis C at this stage, so we have a strong need for better therapies.
Henrik - There's absolutely a very strong need for new therapies.
Chris - And what have you done?
Henrik - So what we've done is we've taken a non-traditional approach. Rather than trying to attack the virus directly, we're attacking it indirectly by sequestering a host factor that the virus uses for its replication. It turns out that when we do it, we get a drug that is very potent in the chimpanzee model, which is the only other species (other than humans) that can contract HCV, so it packs a combination of very good potency and good safety, and a unique barrier to resistance.
Chris - So first of all, tell us, what is the new drug and how does it work?
Henrik - It works by binding to and sequestering an endogenous microRNA called microRNA-122 that is specifically expressed in the liver and which the virus uses for its replicative cycle. And sequestering this basically removes it from the virus and hence stops the virus replicating.
Chris - Why should the virus rely on a human cellular factor, this microRNA to grow at all? Why does it need that?
Henrik - Well, viruses depend on a lot of host factors. They do not encode all of the functions they need to complete their life cycles. So, when they enter cells, they do co-opt a lot of different host factors to complete that.
Chris - And your new agent, how does it work? What does it do to that microRNA in the liver cells to make it so that the cells will no longer allow the hepatitis C to grow there?
Henrik - Well, the microRNA in the infected liver cell basically binds to two sites in the 5' end of the HCV genome. And although the mechanism by which this binding facilitates replication is not entirely known in details at this point, it is known to be a direct binding event between the microRNA and the HCV genome.
Chris - So in some way, that microRNA encourages the virus. It then enables the virus to copy its genetic material.
Henrik - Yes. So, what our drug does is it binds competitively to the microRNA and sequesters it in a form that it can no longer bind to the HCV genome.
Chris - Where else in the body would your cells make microRNA-122, this particular linchpin, and does your drug therefore have the potential to inactivate a key component of cells in other bits of the body and therefore, cause side effects?
Henrik - All present data shows us that microRNA-122 is a liver-specific microRNA and it's normal function is involved in the biosynthesis and metabolism of lipids and cholesterol. So, what we observed as the only other effect so far in extensive toxin pharmacology studies when we inhibit the microRNA-122 is the expected reduction in plasma levels of cholesterol.
Chris - And so, when you inhibit this particular microRNA in the liver with your drug, what happens to the hepatitis C infected chimpanzees you were trying it on?
Henrik - So we injected them once weekly every 12 weeks and during the whole dosage periods, we saw a steady decrease of virus titres, both in plasma and in the liver. And at the end of dosing, this effect lasted a couple of months post-dosing, consistent with the fairly long half-life of the drug. So, over a period of 5 months where we kept the microRNA fully suppressed in these chimps, they was a steady decline and very strong response on the virus that did not bounce back at any point in time. I think this combination of a good response in the virus and the safe treatment, combined with the apparent complete absence of a viral breakthrough through this extended period of time, that's the combination that really creates excitement in the community.
Chris - And the next step is presumably now to try this in humans?
Henrik - Yes. We've so far conducted the first study in healthy volunteers. It's a single-dose study, single ascending dose study. We're currently conducting a multiple ascending dose study to define the dose and the dosing schedules and we'll hopefully move to patients in the near future. We haven't quite worked out the design and where those studies are going to be conducted, but we will setup a patient information centre on our homepage where patients interested in this new drug can get the relevant information.
Chris - And you can get more relevant information in the journal Science this week and the drug that Henrik was talking about is SPC3649. That was Dr. Henrik Øren who is from Santaris Pharma.
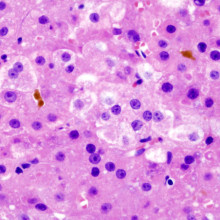
20:12 - Hepatitis C Virology
Hepatitis C Virology
with Dr Joe Grove, Birmingham University
Chris - With us this week is Dr. Joe Grove who is a hepatitis researcher. He's from Birmingham University and he works on the way that hepatitis C viruses actually get into our cells and then escape from the immune system. One of the things about hepatitis C is once you've got it, in the majority of cases, you don't actually get rid of it again. It stays with you, which means it must be able to get away from the immune system. Joe, how are you?
Joe - Hi. How are you?
Chris - Very well. Welcome to The Naked Scientists. So tell us a little bit first of all about what actually is hepatitis C.
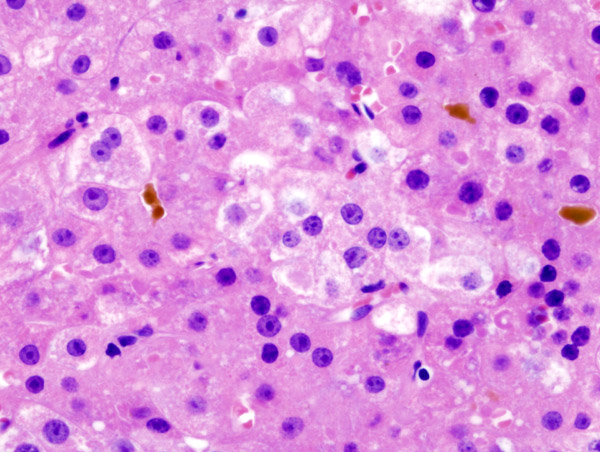 Joe - Okay. It's quite a common misconception that hepatitis C is some way related to the other hepatitis viruses. So it's good to start and establish that it isn't. The word hepatitis is derived from the latin for liver. So the hepatitis viruses share one thing in common. They infect the liver. However, in the same way that you could describe a crab, a dolphin and a jelly fish as ocean creatures, these viruses are completely different but they just share a habitat. With hepatitis C, if you look at its closely related viruses, they are viruses such as dengue virus which transmits between humans via mosquitoes, and there's also pestiviruses which are related that infect cattle. Hepatitis C seems to be quite the black sheep of the family within this group and it's quite unusual in that it can only be found in humans. Although as Henrik eluded to earlier, you can get infection of chimpanzees as well.
Joe - Okay. It's quite a common misconception that hepatitis C is some way related to the other hepatitis viruses. So it's good to start and establish that it isn't. The word hepatitis is derived from the latin for liver. So the hepatitis viruses share one thing in common. They infect the liver. However, in the same way that you could describe a crab, a dolphin and a jelly fish as ocean creatures, these viruses are completely different but they just share a habitat. With hepatitis C, if you look at its closely related viruses, they are viruses such as dengue virus which transmits between humans via mosquitoes, and there's also pestiviruses which are related that infect cattle. Hepatitis C seems to be quite the black sheep of the family within this group and it's quite unusual in that it can only be found in humans. Although as Henrik eluded to earlier, you can get infection of chimpanzees as well.
Chris - How does it spread in the majority of cases?
Joe - It's a blood-borne virus and we only first isolated the virus in the last 20 years and that's allowed research to start. Before that, we didn't know what caused this form of hepatitis and it was actually transmitting, certainly in the western world, predominantly through blood transfusions. Much the same way that HIV was in the '60s and '70s. However, since we've discovered what the virus is, we can test blood so there's no danger from blood products, certainly in the western world. And now, the predominant route of transmission in the western world is via intravenous drug users sharing needles. However, in the developing world, there is a problem with poor medical practices and poor sterilization of medical implements, leading to transmission. So for instance, in parts of Egypt, you have a very high level of infection because of a vaccination campaign where the needles weren't sterilized properly, and as a result, 40% of the population contracted hepatitis C.
Chris - Yes, I think that the current numbers are 1 person in 10, if you just pick a person at random, has actually got it. Does the virus then just home in on the liver or does it affect other tissues too?
Joe - Well, there is some evidence to suggest that the virus may have reservoirs throughout the rest of the body but the predominant site of replication is within hepatocytes. These are the cells that function within the liver. So, once the virus enters the bloodstream, it will circulate in the blood, interact with a specific receptors expressed on hepatocytes and enter liver cells. This is the place where it replicates. Viruses are one of the most simple forms of life and they are completely dependent on 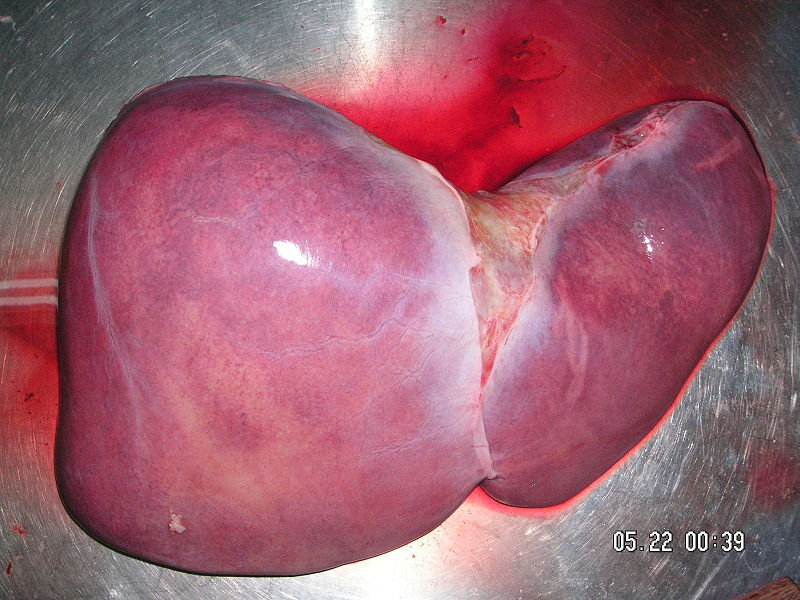 their host. So the entry of a virus into its host cell is a prerequisite for its replication.
their host. So the entry of a virus into its host cell is a prerequisite for its replication.
Chris - So in other words, it gets into the liver cell, hijacks it, turns it into a hepatitis C factory, and then that hepatitis C infected cell just makes more copies of hepatitis C which then goes around the bloodstream, infects other liver cells, but can also - if someone sticks needle in there, and then shares that needle, infect another person. But the key thing is, there are many different types of hepatitis virus, but they don't cause an infection for life. But hepatitis C does. So, why is that?
Joe - You're right. It's peculiar in this sense. It seems particularly able to evade the immune system. There seems to be lots of reasons for this namely - to start with, hepatitis C is what is known as an RNA virus. That is, it's genetic material is made of a chemical related to DNA, called RNA. However, this RNA has a higher mutation rate. Therefore, the virus can change more quickly than a DNA organism. And this allows it to stay one step ahead. It's continually evolving to evade the human immune system. However, it also seems to be able to perform other tricks. For instance - the lab I work in with Professor Jane McKeating in Birmingham, we've been looking at some of the ways that the virus may evade the immune system. The virus can interact with B cells and these are cells of the immune system, and it seems like it can be transmitted within these B cells. So it hides within them as kind of a 'Trojan horse' model of transmission. And also, some of the work we're doing would suggest that the virus can transmit directly between cells, avoiding the immune system that's in the bloodstream.
Chris - So although, if you look at people's blood who have hepatitis C, you can find lots and lots of antibodies against HVC, because the virus may not necessarily be in the same blood space as those antibodies, they can't actually neutralize it?
Joe - Yeah. It's very complicated. We can take the blood of infected individuals and take the antibodies from these individuals. And they seem to show activity in the lab. We can show that we can stop a virus in the lab. However in that patient, that antibody doesn't help them in any way. It may help control the virus, but it doesn't help them resolve the virus. So we are particularly interested in how this is achieved and in particular, we're looking at the entry of the virus, and this is the stage at which neutralizing antibodies act. So a neutralizing antibody will stick to the surface of hepatitis C, and in doing so, inactivate the glycoproteins which are the proteins that sit on the virus and interact with the receptors that are on the cell.
Chris - So the point we're getting to is what we need if we want to protect people is we've got to have antibodies in the bloodstream before someone gets infected, so that they can interrupt that process upstream of the virus actually getting into cells because once it does, that is probably too late then.
Joe - Yes, correct. It's important that we identify which regions of the virus are important for the entry into the cell. So, you will raise antibodies against lots of different parts of the virus, but some of those are decoys. Some of those are red herrings so that the immune system follows this particular part of the virus and it won't help them at all. So, we need to identify which regions of the virus are important for neutralization as well.

What causes Red Eye in Photographs?
Red eye occurs because when you take a flash photograph - the camera produces a big burst of light to illuminate the subject and that burst of light goes in through the open pupil of the subject, and bounces off the back of the eye.
There's a layer called the choroid which has a very rich blood supply inside the eyeball and that reflects red light, the colour of the blood, back out of the front of the eye and into the camera.
This happens so quickly that there's not time for the pupil to contract before the camera takes the picture, so you see this red reflection of the interior of the person's eyeball.
Red eye reduction works by the camera shining a brightish light or doing a few "spoof" flashes at the subject first.
This constricts the pupil down in the person you're taking a picture of, and then it takes the proper picture. This means that there's a very small aperture in the front of the eye as the pupil has got very tiny, therefore much less light gets in and it's much harder for light to reflect back out again. And that's how red eye reduction occurs.
Why are some people more prone than others? Perhaps, the pictures that you've been taken in are not pictures involving flash photography, so you don't seem to have the effect. Perhaps also, you're not directly in line with the camera flash.
If the flash didn't actually illuminate the eyeball sufficiently on its interior, not enough light will come back out of the camera. One other possibility is that you have very, very dense pigment epithelium. This is a layer of melanin (the same stuff that gives you a sun tan) inside the eye and that's what soaks up some of the extra light.
People also naturally differ in how wide their pupil is in any particular light. People with wider pupils are more likely to end up with red eye in photos, whereas those with smaller pupils are less likely.

30:33 - Safer Blood Transfusions
Safer Blood Transfusions
with Jean-Pierre Allain, University of Cambridge & Lorna Williamson, NHS Blood and Transplants
Meera - Until 1991, blood transfusions were a common cause of hepatitis C infection, as the virus is spread by blood to blood contact. Famous figures such as the late Anita Roddick, founder of The Body Shop and stuntman, Evel Knievel were believed to have contracted hepatitis C in this way. But since 1991, the virus has been screened for in all blood donations in the UK. So just how do our blood services screen for not only this virus but other infectious agents as well? And as a result, how safe is our blood? To  find out, I spoke to Jean-Pierre Allain, Professor of Transfusion Medicine at the University of Cambridge.
find out, I spoke to Jean-Pierre Allain, Professor of Transfusion Medicine at the University of Cambridge.
Jean-Pierre - Actually, we screen essentially for viruses and there are three main viruses we are screening for. The first one and most well-known is HIV, the second is hepatitis B virus or HBV, and the third one is hepatitis C virus or HCV.
Meera - How do you actually go about testing for these blood samples? So, somebody comes in to give a donation, what happens?
Jean-Pierre - First, we ask the potential donors to fill a questionnaire regarding potential infections and we also ask questions about their sexual behaviours because it has to do with the higher risk of infections, and also about drug abuse. Then we collect two tubes of blood, one with which we test for evidence of viral infection, essentially antibodies to HIV and HCV, and for hepatitis B surface antigen. In addition to that, with the second tube, we test for the genome of the virus. In other words, a direct gene or nucleic acid of the virus.
Meera - How do you actually go about looking for antibodies in somebody's blood and what does this tell you then about their infections?
Jean-Pierre - When we test for antibodies, we are looking to determine whether the individual has been in contact with the virus or not. We detect these antibodies by capturing the antibodies on a range of antigens from the virus itself. If the antibodies are present, they are binding to these antigens and then we detect the presence of antibodies.
Meera - What then happens in the next step where you start looking at the genomes of these viruses?
Jean-Pierre - Every blood sample is tested for this genome. Actually, not individually like we do for antibodies, but in what we call pools of samples from the donors - at the moment, this pool is 24. When we test for the genome in these pools of 24 and we find a positive reaction, then we go back to the individual donations to identify which donor is responsible for the pool being positive. Then we correlate that with the antibody being found separa tely.
tely.
Meera - Now I imagine when looking for particular genomes of the virus, it must be quite difficult because viruses evolve very rapidly. So, which part of the genome do you look at?
Jean-Pierre - The virus itself has a tendency to easily modify its genome. So eventually, it could be difficult to detect. Except that we choose an area of the genome we know is what we call conserved which mean that it doesn't change at all or very little.
Meera - Why is this area conserved?
Jean-Pierre - Because it's functionally critical. And the more critical it's functionally for the virus to replicate, the more it needs to stay the same.
Meera - Now, having done all of these tests and looking into the blood, what is the risk of somebody now who receives a blood transfusion receiving infected blood?
Jean-Pierre - If we talk about the hepatitis C virus and HIV, the risk is calculated at about 1 in 3-5 million which means that it's less likely to be infected than being hit by lightning if you stroll on a Sunday afternoon.
Meera - Jean-Pierre Allain from the University of Cambridge. So we now know how we screen for viruses that we know of and know the biology of, but what about infections that we haven't met before? What about the unknowns? I asked Lorna Williamson, Medical and Research Director for NHS Blood and Transplants.
 Lorna - Virtually every blood donation that's collected is separated into different parts. When people say blood transfusion, they generally mean the red blood cells which are given to carry oxygen and treat bleeding and severe anaemia. But we also can take the plasma, the liquid part of blood, and that is used to replace clotting factors in patients who are bleeding, and then finally, we can make blood platelets which are little discs that circulate in the blood and they are also important to prevent bleeding. We try as hard as possible to remove all the parts of blood that won't benefit the patient. We filter out the white blood cells which, in a transfusion, don't offer any benefit. If the patient needs red cells, we will take away all the plasma and we can give the patients exactly what they need and no other part of the blood. And then where we can, we would alter the blood donation in some way to try and kill or remove any viruses or infectious agents. So for example, plasma can be heat treated or treated with chemicals and there are new methods coming through to treat platelets with chemicals to kill viruses and bacteria.
Lorna - Virtually every blood donation that's collected is separated into different parts. When people say blood transfusion, they generally mean the red blood cells which are given to carry oxygen and treat bleeding and severe anaemia. But we also can take the plasma, the liquid part of blood, and that is used to replace clotting factors in patients who are bleeding, and then finally, we can make blood platelets which are little discs that circulate in the blood and they are also important to prevent bleeding. We try as hard as possible to remove all the parts of blood that won't benefit the patient. We filter out the white blood cells which, in a transfusion, don't offer any benefit. If the patient needs red cells, we will take away all the plasma and we can give the patients exactly what they need and no other part of the blood. And then where we can, we would alter the blood donation in some way to try and kill or remove any viruses or infectious agents. So for example, plasma can be heat treated or treated with chemicals and there are new methods coming through to treat platelets with chemicals to kill viruses and bacteria.
Meera - How does treating them with heat help make them safer and remove any viruses?
Lorna - Well it simply prevents the virus genome from replicating. It destroys the ability of the DNA and RNA to copy itself, so the virus can't multiply.
Meera - As well as heat treatment. What other precautions that are taken?
Lorna - What we would really like would be a method of either heating or treating with chemicals the red blood transfusions which are the biggest type of blood product that's used and such methods are in development. The one thing we are assessing for blood transfusions are new filters to remove infectious agents called 'prions.' They're small bits of protein that cause prion diseases like variant CJD and BSE in cattle.
Meera - And what do you think, just lastly then, the kind of risks are of any infection or viruses that you just really aren't aware of at the moment and how prepared do you think the system is to deal with this?
Lorna - So we work very closely with the Health Protection Agency where the monitoring systems for infections that may come to this country from elsewhere in the world. Swine flu is a good example of that. There are also systems for monitoring infections in farm animals and we work very closely with other blood services throughout the world. With migration and international travel, infectious agents can move around the world very rapidly. So, it is a case of maintaining high vigilance on an international level.
38:47 - Hepatitis C in the Clinic
Hepatitis C in the Clinic
with Dr Graeme Alexander, Addenbrookes Hospital
Kat - What does Hepatitis Cactually do to the body, and how can we treat it? It's now time to find out from Dr. Graeme Alexander, he's a Clinical Hepatologist at Addenbrooke's Hospital. So, hello Graeme. Welcome to the show.
Graeme - Hello.
Kat - Now, just before we start, we've had a quick question from SecondLife, from Dali Waverider. He wants to know how many classes of hepatitis virus are there and how are they different?
Graeme - Well a lot of viruses can affect the liver, but there's five that we recognize as important in the liver and they're Hepatitis A, B, C, D, and E - rather imaginative aren't we?
Kat - Fair enough.
 Graeme - The most important thing about hepatitis C is that it, by and large causes an infection that lasts for a lifetime while the other viruses don't (usually). So that differentiates it quite easily. And it's an RNA virus whereas hepatitis B is a DNA virus infection. But that's a semantic difference. The important thing about hepatitis C is that it causes lifelong infection in many people.
Graeme - The most important thing about hepatitis C is that it, by and large causes an infection that lasts for a lifetime while the other viruses don't (usually). So that differentiates it quite easily. And it's an RNA virus whereas hepatitis B is a DNA virus infection. But that's a semantic difference. The important thing about hepatitis C is that it causes lifelong infection in many people.
Kat - So let's look at this kind of infection. So we've discovered that it goes in to the cells in the liver and it sort of turns them into little virus factories. What effect does that actually have on the liver?
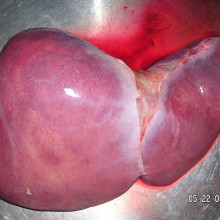
Graeme - Well I think the most important thing to stress straight away is that for most people, you don't know you've got hepatitis. It doesn't cause liver damage in the majority of people. But about 5%, perhaps as high as 20%, of people in some populations get into a situation where they develop liver damage. And you might not know you've got liver damage evolving until maybe 50 or 20 years after you've been infected with the virus. In a way, the virus wears the liver down over a period of years, causing scarring within the liver and once the scarring is present, then the patient is at risk of liver failure and the complications thereof.
 Kat - So presumably, there must be something to do with the differences between people, as to why some people are susceptible to having liver damage from hepatitis infection and why some people aren't. Do we know anything about those kind of genetic factors?
Kat - So presumably, there must be something to do with the differences between people, as to why some people are susceptible to having liver damage from hepatitis infection and why some people aren't. Do we know anything about those kind of genetic factors?
Graeme - Well, we do. We know quite a lot about those genetic factors and we know that for example, men do much worse than women. We know that it matters how old you are when you catch the virus. Men who are over 40 when they catch the hepatitis C do very much worse than say, a young girl of 20 who catches the virus. We know that people who are overweight do very much worse than the patients who are thin, and we know that people who drink heavily increase their risk of liver damage from hepatitis C very, very much so.
Kat - And this, we bring in a little question here from Meera Maquana who says, "Why does chronic liver disease, and things like chronic drinking, lead to cirrhosis and what's the process of this scarring that's going on?"
Graeme - The liver cells are part of a complicated organ - the liver - where there's lots of different types of cells. There's another cell sitting next to the liver cell called a stellate cell and there are signals sent out by an injured liver cell to those stellate cells which scar the liver. So the liver ends up being scarred in exactly the same way as someone who's had an operation and there's a scar on their skin, but this scar is spread finely and diffusely throughout the whole liver.
Kat - Now, something I do know from my work at Cancer Research UK is that rates of liver cancer are going up and we know also that liver cancer is linked to hepatitis infection. How does that work? What's the link there?
Graeme - Well it's all through liver disease. All forms of liver disease predispose 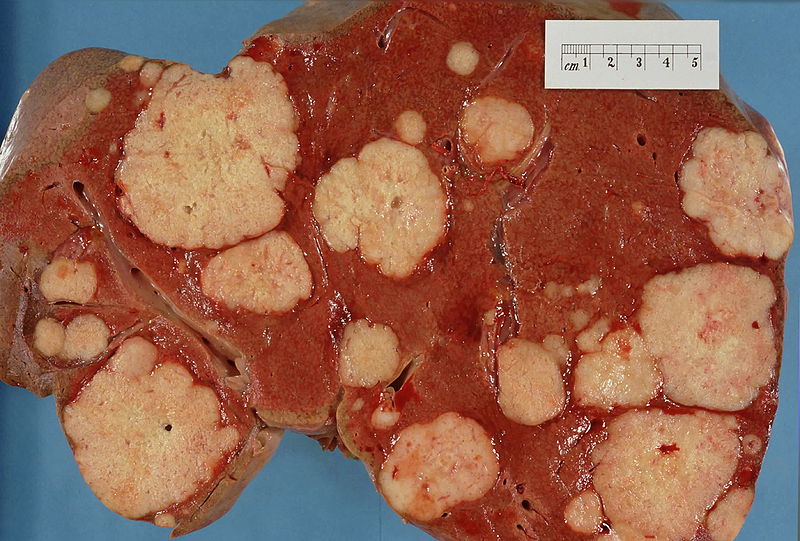 patients to liver cancer in the longer term and it's one of the things that we take great care for is to look for liver cancer at an early state to try and pick it up. Hepatitis C is one of the major causes of liver disease in this country. So it's now one of the major causes of liver cancer. And if you have advanced liver disease, you've got a one in four risk of getting on to get liver cancer subsequently. It's also a significant problem.
patients to liver cancer in the longer term and it's one of the things that we take great care for is to look for liver cancer at an early state to try and pick it up. Hepatitis C is one of the major causes of liver disease in this country. So it's now one of the major causes of liver cancer. And if you have advanced liver disease, you've got a one in four risk of getting on to get liver cancer subsequently. It's also a significant problem.
Kat - So, it strikes me. Is that it's fairly important to try and treat, and probably even prevent hepatitis infection. Where are we currently with treatments? And we've heard already about some current ideas, you know, this microRNA drug for future treatments, but where are we currently with treating hepatitis?
Graeme - Well, there's been enormous progress in the last four or five years. We've been known about interferon which is what you and I produce when we get an infection, flu for example. We've known about that for quite a time and on its own, it didn't really work very well. And then another drug called 'ribavirin' was introduced, but that on its own didn't do very well either. But when we combined these two drugs, we got striking improvements in response rates which was unexpected, but very gratifying. So now, we think that we can treat something towards about a half of the patients that come to see us. But the most important point to stress here is that the earlier we treat the patients, the better, so younger patients did much better with treatment than older patients. So it's quite important that we do see people at an early stage and we can consider treatment as early as possible in the course of the disease.
Kat - And do we already have vaccines against hepatitis C? What hope is there for preventing the transmission of it?
Graeme - Well, hope is all we have at the present. There are a lot of people working on this area and you've heard from Joe Grove earlier about the virus evading the immune responses and the number of tricks that it has to do this. The problem we face at the present is we don't recognize the population who are immune to hepatitis C in the long term. They don't develop neutralizing immunity and if you can't develop neutralizing immunity, it's very hard to see how you would target a vaccine response. So sadly, I think, vaccine responses are some distance away and we're really looking at prevention for now rather than a vaccine.
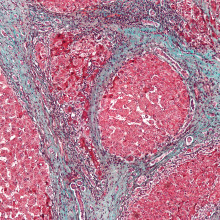
How does liver disease lead to cirrhosis?
We put this to consultant hepatologist Graeme Alexander:
Graeme - The liver cells are part of a complicated organ - the liver - where there's lots of different types of cells. One cell type sitting next to the liver cell (the hepatocyte) is called a stellate cell, and there are signals sent out by an injured liver cell to these stellate cells, which then scar the liver. So the liver ends up being scarred in exactly the same way as someone who's had an operation and there's a scar on their skin, but this scar is spread finely and diffusely throughout the whole liver.

Can Hepatitis B be transmitted in urine?
We put this to consultant hepatologist Graeme Alexander, from Addenbrooke's Hospital, Cambridge...
Graeme - When I meet patients in the clinic, I get two types of question. One, from a patient who's scared that they'll transmit hepatitis B to someone they love, and then the other, from the person that they love, wondering if they'll catch it from their partner.
And the answer is, that you can't catch hepatitis B from urine. These viruses are actually quite difficult to catch, both hepatitis B (and another blood-borne virus called hepatitis C).
Hepatitis B is acquired largely by contact with blood or through sex, and hepatitis C pretty well only by contact through blood.
So, you can live with someone for many, many years and not catch hepatitis B or C from them because close contact in the family situation is entirely safe.
Chris - And I think the other mitigation is that there is a good vaccine for hep-B, isn't there? So if we identify people in a family situation who have one carrier and one person who isn't infected, we can vaccinate the uninfected person to protect them.
Graeme - It's probably the most effective vaccine that we've ever come across, very effective. And very safe once you've been vaccinated.
Does money carry germs?
Yes, certainly money can be a vector for infection and as an example, lets look at norovirus, which causes winter vomiting disease (but more recently, vomiting all around the world at all times of the year - it's becoming incredibly common). Norovirus is a tiny particle, one 30,000th of a millimetre across, can very easily be transmitted from one surface to another. They can survive seven changes - so if you touch something, and someone else touches it and picks it up, they can then transfer it seven times and the virus still remains infectious. You only need to pick up one of them to get infected. Yuck!

54:14 - How do farmers propagate seedless crops?
How do farmers propagate seedless crops?
We put this question to Stephen Tomkins, from Homerton College, Cambridge, and Jennifer Schultz Nelson, Horticulture Educator with the University of Illinois:
Stephen - You need to know something about why they have no seeds and that's because they're often triploids. That is, they've got three sets of chromosomes and you and me have two. And when it comes to sexual cell divisions in meiosis, they can't organize their chromosomes properly and they fail, and therefore, they have no babies.
So the question that you want answered is how do you get to a triploid fruit? And the answer to that is a little bit complicated. Most plants have two sets of genes, two sets of chromosomes and they divide equally when cell divisions take place. But plants are very tolerant of having multiple sets and that's called polyploidy.
Polyploid plants can be triploid, tetraploid, pentaploid, and up the numbers go! And it doesn't often badly affect the viability of the plant. They grow. They photosynthesise. They can produce fruits. But if they have odd numbers of chromosomes, they are stuffed. They can't have any babies at all. And that happens to the triploids and the pentaploids because you can't divide odd numbers evenly.
If you want to multiply up that plant, you can do it by cloning or vegetative propagation. And that's an important thing for people setting out to grow large orchards of triploid fruit.
Diana - So you can propagate plants which are gameteless so they make seedless fruit. And here's a little more on how it's done.
Jennifer - When humans get involved, they try to manipulate things and they can do things like treat a plant with a chemical called 'colchicine' which disrupts the meiotic process. So when the plant is producing pollen grains and ovules, they will produce a gamete that has double the amount of chromosomes. So then when it is pollinated by the original plant that provides one copy of chromosomes, the result is a triploid plant.
We talk about cloning being a really modern method, but actually, horticulturists have been using it for quite some time. Any time you're taking a cutting of a plant and rooting it using different plant hormones or simply sticking it in a glass of water, watching the roots grow and then planting that plant, that's cloning, also called 'vegetative propagation.'
I actually found some information that when you talk about polyploidy in plants and it sounds like a really rare thing, but it's actually not that rare. Scientists think that anywhere from 30 to 70% of angiosperms from the plant kingdom are polyploidy and polyploidy plants tend to be larger. And so, maybe they're more competitive in nature and so that might be some advantage for that to be selected for in nature.
Diana - Triploid plants can be reproduced using cloning and vegetative propagation as well as chemical mutagens like 'colchicine.' But many farms will grow the fertile parent plants nearby so that more seedless offspring can be produced later.











Comments
Add a comment