In this week's NewsFlash, we discover how storms create slow earthquakes and how a local star, Betelgeuse, could explode very soon. We also hear of an accurate way to date pottery and explore the physics of helicopter seeds. Plus, this week in Science History saw the death of John Logie Baird, pioneer of television.
In this episode

Typhoons trigger earthquakes
Scientists have uncovered evidence that large storms can trigger certain types of earthquake.
 Writing in this week's Nature, Taiwan-based researcher ChiChing Liu from Academic Sinica in Taipei together with two scientists from the US, explains how between 2002 and 2007 he and his colleagues used underground strain-sensitive devices to follow how the ground deformed over time.
Writing in this week's Nature, Taiwan-based researcher ChiChing Liu from Academic Sinica in Taipei together with two scientists from the US, explains how between 2002 and 2007 he and his colleagues used underground strain-sensitive devices to follow how the ground deformed over time.
The researchers found that they could pick up the arrival of typhoons, which occur predominantly in the second half of each year. These storms are accompanied by very low pressure, which usually causes the ground to swell and this is what the team could see on their subterranean strain-meters. But occasionally they would pick up the reverse - the ground appeared to have shrunk.
They detected eleven events like this, all associated with typhoons. The likelihood of this occurring by chance is less than one in a million, and the only explanation, say the scientists, is that the typhoons are occasionally triggering 'slow earthquakes', which are ground movements that occur over much longer timescales - hours to days - compared with their normal vigorous counterparts.
The storms unleash the slow quakes, say the researchers, by increasing the stress across faults. This occurs because the arrival of the storm causes atmospheric pressure to drop over land, but to remain unchanged over the sea. This stretches the fault and if it is primed to move then a quake follows.
Paradoxically, this mechanism might actually help to protect Taiwan, where Philippine Sea plate and the Eurasian plate are running into one another at more than 8cm per year, by helping to periodically 'unload' the fault and preventing the build-up of energy that would otherwise be unleashed subsequently and with potentially deadly effect.

Betelgeuse the shrinking star
Betelgeuse, as well as being an 80's classic film is one of the brightest stars in the sky. It is also Orion's right shoulder. It is one of the largest stars we know known as a red supergiant, with a mass about 20 times larger than the sun and a radius about 1000 times larger than the sun.
 This week Charles Townes from Berkley in California has announced that its radius has shrunk by about 15% over the last 15 years and this contraction is getting faster.
This week Charles Townes from Berkley in California has announced that its radius has shrunk by about 15% over the last 15 years and this contraction is getting faster.
This is interesting because Betelgeuse is already in the closing stages of its life despite only being about 8-9 million years old where as our sun is about 5 billion years old.
It is so large that its gravity crushes the gasses in its cores to much higher pressure and temperature than our sun. This speeds up the nuclear fusion reactions going on and means that Betelgeuse has already burned up all its hydrogen, When this happens to a star its core cools down, and as the only thing which supports the star against gravity is gas pressure, the core starts to collapse, as it does this it heats up until it can start burning helium to form carbon.
Depending on the size of the star this process can occur several times, each time the core gets smaller and hotter, and starts burning heavier elements. first it burns Hydrogen then Helium, to form Carbon, and this reacts to from Neon then oxygen and Silicon. At each stage the nuclear reactions release less energy until there is nothing left to burn.
The change in size of Betelgeuse indicates that something interesting is happening in the core.
Why is this interesting to anyone other than Astronomers? Well in a large star like Betelgeuse, eventually there are no more reactions to take place, and the core keeps shrinking until it effectively forms a huge atomic nucleus - a neutron star. This releases an immense amount of gravitational energy (about 1/4 of E=mc2) and the star explodes incredibly violently as a supernova.
If Betelgeuse goes supernova it will be as bright as the full moon but concentrated into a point, and it will be visible during the day for several months. It is 600 light years away so although it would be close enough to be spectacular, it should be distant enough to be safe for the planet earth.
Whether it will actually go bang in the next few years, nobody knows, as we have never watched the early stages of a supernova, as they are so rare, and noone has been lucky enough to have a telescope pointing at the right star at the right time, but if it does it should be spectacular.
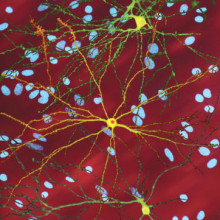
Understanding Huntington's Disease
Huntington's disease is a degenerative disease of the nervous system that sets in when a person is in their 30s or 40s, although they show no signs of the disease before it kicks in. Over a decade ago, researchers discovered that sufferers all have a fault in a specific gene, which makes a protein called huntingtin.
 But why are faults in this protein not harmful when sufferers are young, but has serious effects when they hit later life? That's what researchers at the University of Illinois wanted to answer. And in a new paper published in the journal Nature Neuroscience this week, the team, led by Scott Brady, may have discovered how huntingtin wreaks its havoc on the nervous system.
But why are faults in this protein not harmful when sufferers are young, but has serious effects when they hit later life? That's what researchers at the University of Illinois wanted to answer. And in a new paper published in the journal Nature Neuroscience this week, the team, led by Scott Brady, may have discovered how huntingtin wreaks its havoc on the nervous system.
The scientists discovered that the faulty version of huntingtin, found in patients, switches on an enzyme called JNK3, which is only switched on in nerve cells. At low levels of huntingtin, this activation of JNK3 blocks transport within nerve cells, stopping nerve cells from shuttling proteins from the middle of the cell along long fibres called axons.
This is bad news for nerves, as it means signals don't get properly transmitted down the nerve fibres, which causes the nerves cells to eventually die off, causing the problems of Huntington's disease.
The scientists think that activating JNK3 cuts down on transport in the nerve cells, but doesn't completely stop it. When nerve cells are young, they can cope with a reduction in transport. But as a person gets older - and their nerve cells get older - the cells become less able to cope.
The researchers think that this pattern of progressive nerve breakdown could also play a part in diseases like Alzheimer's, and other adult-onset neurodegenerative diseases. In fact, the scientists have coined the term 'dysferopathy' to describe these kinds of diseases - the word is from the Greek 'fero', meaning transport. So perhaps by targeting this "Achilles heel" of transport within nerve cells, we might be able to find new ways to prevent or treat these kinds of disorders in the future.

Plants take a leaf out of insects' books
Scientists have discovered the trick that keeps certain trees' seeds aloft - and it turns out they use the same strategy as insects.
 Writing in this week's Science, Harvard researcher David Lentink and Caltech scientist Michael Dickinson explain how they have cracked the puzzle of how the mini 'helicopter' shaped seeds of maples and hornbeams manage to fly so well.
Writing in this week's Science, Harvard researcher David Lentink and Caltech scientist Michael Dickinson explain how they have cracked the puzzle of how the mini 'helicopter' shaped seeds of maples and hornbeams manage to fly so well.
"We immersed in oil a robot fly designed to mimic the seeds' wing shape and programmed it to turn in a circular fashion resembling the seeds as they fall," explains Michael Dickinson. "By illuminating a slice at a time of the oil with a laser beam we could see how the fluid was flowing around the wings."
What the team saw was a phenomenon called a leading edge vortex - LEV - which is like a miniature tornado turned on its side and sitting against the wing. "These vortices create enormous lift, which is what keeps the seeds up for much longer as they fall," Dickinson explains.
To prove that real seeds do what the robot model predicts they should, Lentink came up with a way to film the seeds 'falling' in a wind tunnel. The vortices were clearly visible and, intriguingly, they match the exact same mechanism that keeps insects aloft.According to Dickinson, "the falling seeds, as well as insects (which engineers say shouldn't be able to fly!), have their wings arranged with a very high angle of attack, which is what makes these vortices. Insects twirl their wings like a figure of eight in flight, these seeds spin."
The result is an incredible example of convergent evolution - how two totally different organisms have hit upon the same solution to a problem - but can it inform future flight? "We think it might help us to solve some problems regarding how to build better turbine blades in future," says Dickinson.

Pot Dating
If you are an archeologist looking at a new site, one of the first things you want to know is how old it is. Radiocarbon dating can answer this question for organic objects that contain carbon, but carbon can be quite rare as organic material gets eaten.
One thing that is very common in almost all archeological sites is pottery. It is easy to make and cheap, it breaks easily and you can't recycle it, so there was a lot thrown away and it lasts thousands if not millions of years. So it gets everywhere, unfortunately there is no carbon in it and it is very difficult to date.
However Moira Wilson and colleagues may have come up with a solution. When you make pottery, you fire it. You heat clay up to a temperature between 1000 and 1400 Celcius which sinters it, causing the particles of the clay to stick together and crucially drives the water out of some of the minerals which make up the clay.
Then as soon as the clay cools down a very slow reaction between these minerals and water starts, and the team is using this reaction to date the pottery.
It is easy to measure the amount of water in the minerals of the pot, you just dry the pot out normally to get rid of the water in amongst the grains, then cook it at 600C for a few hours and measure the difference in weight.
Different pottery takes up water at different rates, but the rate at which it starts taking up water for a couple of days after it is dried out predicts how it will take up water over the next few hundred years very accurately, and crucially as Moira told us:
The reaction is sustained by an incredibly small quantity of water so there's actually sufficient moisture in the atmosphere to keep the reaction going. So it doesn't actually matter whether your brick is sitting on the table, or sitting on the bottom of a lake. As long as there's enough water there to sustain the reaction, any excess water, for example if the material is saturated it doesn't contribute to the reaction it just sits there doing nothing.
In fact the only thing that does affect the rate of uptake of water seems to be the temperature, which is going to be reasonably constant over a fairly large area, so if you can take this into account, by measuring a few pots you know the ages of, you can make remarkably accurate predictions.
They Dated brick from a Charles the second buildig in greenwich which was built between 1664-1669 and altered 1690s as dating from 1691 ± 22 years.
A roman brick was dated as 2000 years old, and is known to be 2001 years old.
They had more problems with a medieval brick from canterbury which they repeatedly dated to be 60 years old. but it turned out that during the blitz there was a major fire in this area, essentially refiring the brick and resetting the timer
If this system turns out to be as good as it promises to be it should be able to date thousands of sites which are so far undated giving us a much more accurate view of the past..
17:59 - A New Element - Ununbium
A New Element - Ununbium
Victoria Gill, BBC Science Correspondent
Chris - And also this week scientists have come up with a reason for you to tear up that periodic table which is on the wall of your chemistry laboratory or your school classroom, and replace it with a new one. This is because we have a new element to add to it. And here to tell us about that new element is someone who occasionally contributes to the Naked Scientists, but is also a BBC science reporter, and that's Victoria Gill.
So, why have we got this new element Victoria?
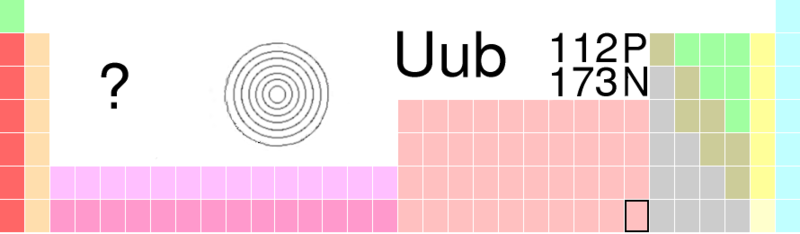 Victoria - Well this is element 112 or Ununbium, called that because its atomic mass, the mass of its nucleus is 112 [from the Latin un, un, bi - one, one, two]. It was discovered by Professor Sigurd Hofmann, in 1996 actually, but it was such a tricky experiment to replicate that it's taken all of this time for the International Union of Pure and Applied Chemistry [IUPAC], which is the official maker and formulator of our ubiquitous and wonderful periodic table to recognise it and credit Hofmann and his team at the Centre for Heavy Ion Research in Darmstadt, Germany, with it's discovery.
Victoria - Well this is element 112 or Ununbium, called that because its atomic mass, the mass of its nucleus is 112 [from the Latin un, un, bi - one, one, two]. It was discovered by Professor Sigurd Hofmann, in 1996 actually, but it was such a tricky experiment to replicate that it's taken all of this time for the International Union of Pure and Applied Chemistry [IUPAC], which is the official maker and formulator of our ubiquitous and wonderful periodic table to recognise it and credit Hofmann and his team at the Centre for Heavy Ion Research in Darmstadt, Germany, with it's discovery.
Chris - How did they actually make the new element, Victoria?
Victoria - So they're using a particle accelerator, and they're essentially firing a beam of ions at a target and fusing two nuclei together. This is a very tricky thing to do when you get to the very heavy elements of the periodic table because these fusion reactions require a lot of energy. To create element 112 or Ununbium as it's temporarily been known, they fired a beam of charged zinc atoms, or zinc ions, at lead atoms in the hope that some of them would fuse together and form a new element, and so they did. What's very tricky about this is that these elements are very unstable; as soon as they form they actually just start to fall apart. The nuclei start to emit energy, but that's quite useful because you can detect the energy that they're emitting and use that to estimate the size of the nucleus. So you can tell that you have a new element. But these fusion reactions don't happen very often, you have to fire this beam at these lead atoms for a very long time and you only get a few successful fusion reactions. In 1996 they only created or saw one atom of element 112. But other teams have had to replicate those experiments in order that IUPAC, the society that draws up our periodic table, can recognise that discovery and say "Yes, this is officially a new element, and we will add it to your periodic table."
Chris - So that's hardly a massive amount of money in the bank in terms of this, four atoms in the last twelve years. But where on the periodic table would we put this, if we were to add the square today, where would we be adding this?
Victoria - Well it's a metal - it would go underneath Mercury on the periodic table, that's where its square would be. In actual fact, because it's been around for so long, because we've known about it for so long other teams have done some experiments on it to find that its properties are very similar to that group and it fits quite nicely into that group.
Chris - Given that it hangs around for such a short space of time, I mean, looking at the half lives of some of the isotopes of element number 112, we're talking less than half a minute, why is this useful?
Victoria - Well this is about really finding out how atoms work and how matter works. And in actual fact what Professor Hofmann's team are doing in the longer term is looking for what they've referred to as "the island of stability". So they think there's a whole new class of elements which have electron shells much further out that are full, that will be able to hang around for much longer, so you're dealing with whole new groups of elements and matter that behaves in a completely different way.
Chris - And given, as you say, that they think there might be the prospects of getting very big elements, built the same way but way beyond the size of this one, could this therefore be used as something like a stepping stone, so you could build some of this and then very quickly add some more to it to get you into the realms of these very big atoms that might have all these exciting properties?
Victoria - That's right, because if, as we're seeing, atoms behave and are built in the way that we would expect, and these fusion reactions are working in the way that we expect, then we can incrementally build these experiments to carry out new fusion reactions and build atoms in exactly the same way, we just need bigger particle accelerators, better equipment and we can get there in the end, it's all just stepping stones as we say.
Chris - Thank you very much Victoria. That's Victoria Gill, explaining how the International Union of Pure and Applied Chemistry, also known as IUPAC for short, have confirmed the existence of a new element this week - was actually developed in the 1990's of course, but had to be proved to exist. They've given it the exciting name of Ununbium temporarily; that's un, un and bi in Latin. But, I'm told the IUPAC, they're going to be considering a new name for it, its official name in the next few weeks. They will listen to what the general public think too. So, if you got a name, you think that this element should have a certain name, tell us what you think and also, tell IUPAC as well.

22:49 - This Week in Science History - John Logie Baird
This Week in Science History - John Logie Baird
Sarah Castor-Perry
This Week in Science History saw, in 1946, the death of John Logie Baird, the pioneer of television. Although his mechanical television system was abandoned in the late 1930s in favour of electronic television, it was still an important period in the 'evolution' of television and some of the techniques still have applications today. Baird continued to be a prominent figure in television technology up until his death.
 Baird was born in 1888 in Scotland. Even as a young man his interest in electrical communication was clear - he used to rig up makeshift telephone exchanges in his bedroom to talk to his friends across the street. He attended university in Glasgow, but unfortunately his degree was cut short by the start of the First World War. Unable to serve in the army because of his health, he became an electrical engineer, then in 1923 decided to move to London to pursue his dream of 'seeing by wireless' using apparatus he had built himself out of old tea chests, hat boxes and a bicycle lantern.
Baird was born in 1888 in Scotland. Even as a young man his interest in electrical communication was clear - he used to rig up makeshift telephone exchanges in his bedroom to talk to his friends across the street. He attended university in Glasgow, but unfortunately his degree was cut short by the start of the First World War. Unable to serve in the army because of his health, he became an electrical engineer, then in 1923 decided to move to London to pursue his dream of 'seeing by wireless' using apparatus he had built himself out of old tea chests, hat boxes and a bicycle lantern.
Viewing moving images on a screen is not the same as viewing people moving about in real life. Television creates an illusion of motion by refreshing the image on the screen line by line so fast that our brains interpret the images as moving.
In his early apparatus, Baird used a version of what is known as a Nipkow disc to help capture the images. This is a disc of metal or cardboard with a series of holes spiralling in to the centre that allow light through a viewing window to a photocell that converts the changing light levels coming through the holes into electrical pulses, a process known as rasterization. These were transmitted along telephone lines to a neon light that was viewed through another Nipkow disc rotating at the same time as the original. The flashing of the light viewed through the rotating disc built up the picture of the object. As the disc rotated, each hole would produce a scan line that together made up the picture. Baird's disc has only 30 holes, meaning that the final image was made up of only 30 lines. In comparison, a modern television that uses cathode ray technology would have around 600 scan lines making up the picture.
 Baird was the first to pass several milestones in the development of television. He made the first transmission of a moving object in 1924 and in 1926 he presented the first transmission of live, moving, monochrome images to members of the Royal Institution and a reporter from the Times in his London laboratory.
Baird was the first to pass several milestones in the development of television. He made the first transmission of a moving object in 1924 and in 1926 he presented the first transmission of live, moving, monochrome images to members of the Royal Institution and a reporter from the Times in his London laboratory.
In 1927 he broadcast the first long distance transmission from London to Glasgow, and in 1928 his assistant Benjamin Clapp travelled with a televisor for receiving the images to New York, where it successfully received a broadcast from Baird in London. He followed this series of incredible breakthroughs later in 1928 with the first transmission of colour images.
 However, Baird was not the only innovator working on television, and he was dealt a blow when the American Philo Farnsworth's electrical television apparatus began to overtake his mechanical methods in terms of quality and ease of use. The BBC, which had been using Baird's system since 1930 decided to switch to electrical television after a successful 6 month trial. The last mechanical television broadcast was made in 1937.
However, Baird was not the only innovator working on television, and he was dealt a blow when the American Philo Farnsworth's electrical television apparatus began to overtake his mechanical methods in terms of quality and ease of use. The BBC, which had been using Baird's system since 1930 decided to switch to electrical television after a successful 6 month trial. The last mechanical television broadcast was made in 1937.
Baird continued to work and innovate in the world of television, patenting systems for both fully electric colour television using cathode ray tubes (still used in non-flat screens today) and 3D television, which he was working on at the time of his death.
Since his death, his legacy has remained. He is recognised as a major figure in the invention of television, something that now plays such a huge role in our lives both as education and entertainment.
Related Content
- Previous Raining Into Space
- Next Your Science Questions










Comments
Add a comment